Abstract
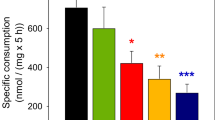
The activities and biotin-dependence of the three mitochondrial biotin-dependent carboxylases: pyruvate carboxylase, propionyl CoA carboxylase, and β-methylcrotonyl CoA carboxylase of primary culture of astrocytes have been examined. An increase of the three mitochondrial carboxylase activities was observed during cell growth, as was the case for developing rat brain. Mitochondrial carboxylase activities from 3-wk-old primary cultures of astrocytes were higher than those in the neonatal rat brain. When astrocytes were grown in a 10% serumenriched medium supplemented with avidin to bind biotin, the mitochondrial carboxylase activities were reduced to 15% of control value. Consistent with these results, after 3 wk in culture, the 3-hydroxyisovaleric acid concentration in the growth medium was tenfold higher than the controls. In this culture condition, cellular growth and the nonbiotin-dependent enzyme, glutamine synthetase, were not modified with respect to control. Primary cultures from newborn rat brain hemispheres are suggested as an experimental approach to the study of biotin deficiency in nervous tissue.
Similar content being viewed by others
Abbreviations
- 3-HIVA:
-
3-Hydroxyisovaleric acid
- GFAP:
-
glial fibrillary acidic protein
- GS:
-
glutamine synthetase
- MCD:
-
multiple carboxylase deficiency
- βMCC:
-
β-methylcrotonyl CoA carboxylase
- PC:
-
pyruvate carboxylase
- PCC:
-
propionyl CoA carboxylase
References
Balazs R., Patel A. J., and Richter D. (1973) Metabolic compartments in the brain: Their properties and relation to morphological structures, inMetabolic Compartmentation in the Brain (Balazs R. and Cremer J. E., eds.), pp. 167–184, Macmillan, London.
Berl S. (1966) Glutamine synthetase determination of its distribution in brain during development.Biochemistry 5, 916–922.
Bock E., Moller M., Nissen C., and Sensenbrenner M. (1977) Glial fibrillary acidic protein in primary astroglial cell cultures derived from newborn rat brain.FEBS Lett. 83, 207–211.
Booher J. and Sensenbrenner M. (1972) Growth and cultivation of dissociated neurons and glial cells from embryonic chick, rat and human brain in flask cultures.Neurobiology 2, 97–105.
Chandler C. S. and Ballard F. J. (1985) Distribution and degradation of biotincontaining carboxylases in human cell lines.Biochem. J. 232, 385–393.
Hansson E., Sellström Å., Persson L. I., and Rönnback L. (1980) Brain primary culture. A characterization.,Brain Res. 188, 233–246.
Hansson E., Rönnbäck, L., Lowenthal A., Noppe M., Alling C., Karlsson B., and Sellström Å. (1982) Brain primary culture—a characterization (part II).Brain Res. 231, 173–183.
Hansson E., Rönnbäck L., Persson L. I., Lowenthal A., Noppe M., Alling C., and Karlsson B. (1984) Cellular composition of primary cultures from cerebral cortex, striatum, hippocampus, brainstem and cerebellum.Brain Res. 300, 9–18.
Hertz L., Juurlink B. H. J., and Szuchet S. (1985) Cell cultures, inHandbook of Neurochemistry, vol. 8 (Lajtha A, ed.), pp. 603–661, Plenum, New York.
Jackson C. W., Brown L. K., Somerville B. C., Lyles S. A., and Look A. T. (1984) Two-color flow cytometric measurement of DNA distributions of rat megakaryocytes in unfixed, unfractionated narrow cell suspensions.Blood 63, 768–778.
Jakobs, C., Sweetman L., Nyhan W. L., and Packman S. (1984) Stable isotope dilution analysis of 3-hydroxyisovaleric acid in amniotic fluid: Contribution to the prenatal diagnosis of inherited disorders of leucine catabolism.J. Inher. Metab. Dis. 7, 15–20.
Kimelberg H. K., Narumi S., and Bourke R. S. (1978) Enzymatic and morphological properties of primary rat brain astrocyte culture, and enzyme development in vivo.Brain Res. 153, 55–57.
Lowry O. H., Rosebrough N. J., Farr A. L., and Randall R. J. (1951) Protein measurement with the Folin phenol reagent.J. Biol. Chem. 193, 265–275.
Norenberg M. D. and Martinez-Hernandez A. (1979) Fine structural localization of glutamine synthetase in astrocytes of rat brain.Brain Res. 161, 303–310.
Sander J. E., Packman S., and Townsend J. (1982) Brain pyruvate carboxylase and the pathophysiology of biotin-dependent diseases.Neurology 32, 878–880.
Schousboe A. (1980) Primary cultures of astrocytes from mammalian brain as a tool in neurochemical research.Cell. Mol. Biol. 26, 505–513.
Schousboe A., Nissen C., Bock E., Sapirstein V. S., Juurlink R. H. J., and Hertz L. (1980) Biochemical development of rodent astrocytes in primary culture, inTissue Culture in Neurobiology (Giacobini E., Vernadakis A. and Shahar A., eds.), pp. 397–409, Raven, New York.
Schrijver J., Dias Th., and Hommes F. A. (1979) Some biochemical observations on biotin deficiency in the rat as a model for human pyruvate carboxylase deficiency.Nutr. Metab. 23, 179–191.
Shank R. P. and Campbell G. L. (1984) α-Ketoglutarate and malate uptake and metabolism by synaptosome: Further evidence for an astrocyte-to-neuron metabolic shuttle.J. Neurochem. 42, 1153–1161.
Shank R. P., Bennett G. S., Freytag S. O., and Campbell G. L. (1985) Pyruvate carboxylase an astrocyte-specific enzyme implicated in the replenishment of amino acid neurotransmitter pools.Brain Res. 329, 364–367.
Suchy S. F. and Wolf b. (1986) Effect of biotin deficiency and supplementation on lipid metabolism in rats: Cholesterol and lipoproteins.Am. J. Clin. Nutr. 43, 831–838.
Suormala T., Wick H., Bonjour J. P., and Baumgartner E. R. (1985) Rapid differential diagnosis of carboxylase deficiencies and evaluation for biotin-responsiveness in a single blood sample.Clin. Chim. Acta 145, 151–162.
Yu A. C. H., Drejer J., Hertz L., and Schousboe A. (1983) Pyruvate carboxylase activity in primary cultures of astrocytes and neurons.J. Neurochem. 41, 1484–1487.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rodríguez-Pombo, P., Sweetman, L. & Ugarte, M. Primary cultures of astrocytes from rat as a model for biotin deficiency in nervous tissue. Molecular and Chemical Neuropathology 16, 33–44 (1992). https://doi.org/10.1007/BF03159959
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03159959