Abstract
Purpose
The volatile anaesthetics enflurane and isoflurane inhibit the endothelium dependent-relaxation in somein vitro preparations. To determine their site of action on the endothelium-derived relaxing factor/nitric oxide (EDRF/NO) pathway, experiments were conducted in a bioassay system.
Method
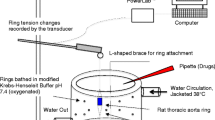
Continuously perfused cultured bovine aortic endothelial cells (BAEC were the source of EDRF/NO while a phenytephrine-precontracted denuded rabbit aortic ring, directly superfused by the BAEC effluent served to detect EDRF/NO. The effect of basal and bradykinin (Bk)-stimulated EDRF/NO release on vascular tension was measured The effect of 4% enflurane or 2% isoflurane on EDRF/NO-induced relaxation was determined.
Results
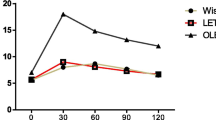
Enflurane added to the perfusate either upstream or downstream in relation to BAEC attenuated the relaxation induced by Bk at low concentrations. On the other hand, isoflurane, added either upstream or downstream to BAEC, potentiated the relaxation induced by the basal release of EDRF but attenuated the relaxation induced by the Bk stimulated release of EDRF Neither enflurane nor isoflurane attenuated the relaxation induced by sodium nitroprusside (SNP), an NO donor.
Conclusion
Enflurane decreases the stability of EDRF/NO released after Bk stimulation while isoflurane can have opposite effects depending on whether the relaxation results from basal or Bk-stimulated release of endothelial derived relaxing factor(s). Isoflurane increases the stability or action of the basal relaxing factor, decreases the stability of the Bk-stimulated relaxing factor (which is probably NO).
Résumé
Objectif
Les anesthésiques volatils enflurane et isolurane inhibent la relaxation dépendante de I’endothélium induite dans des préparations vasculariesin vitro. Des expériences ont été conduites dans un système Bioassay pour déterminer leur site d’action sur la cascade de réaction induite par le facteur relaxant dérivé de I’endothélium/monoxyde d’azote (EDRF/NO).
Méthodic
Des cultures de cellules endothéliales aortiques bovines (CEAB) perfusées en continu constituaient la source de I’EDRF/NO; un anneau aortique de lapin dénudé, préalablement contracté par de la phényléphrine, directement superfusé par I’effluent de CEAB servait à détecter I’EDRF/NO. Nous avons mesuré la relaxation induite sort par la libération basale ou par la libération stimulée par la Bk de facteurs endothéliaux en l’absence et en présence de 4% d’enflurane et de 3% d’isoflurane.
Resultats
Lenflurane ajouté dans le système Bioassay en amont ou en aval relativement aux CEAB atténuait la relaxation induite par Bk a basse concentration. d’autre part. I’lsoflurane ajouté en amont ou en aval des CEAB, potentialisait la relaxation basale de I’EDRF mais atténuait la relaxation induite par la libération d’EDRF provoquée par la stimulation de Bk induite par la libération par la stimulation de Bk. Ni I’enflurane ni l’isoflurane n’atténuait la relaxation induite par le nitroprussiate de sodium (SNP) ou un donneur de NO.
Conclusion
L’enflurane diminue la stabilité de I’EDRF/NO libérée après la stimulation par la Bk; l’isoflurane peut avoir un effet oppose selon que la relaxation résulte de la libération basale ou stimulée par la Bk de I’EDRF. L’isoflurane augmente la stabilite ou I’actrvité du facteur relaxant de base et diminue la stabilité du facteur de relaxation stimulé par le Bk (qui est vraisemblablement le NO).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Vane JR, Änggård EE, Botting RM. Regulatory functions of the vascular endothelium. N Engl J Med 1990; 323: 27–36.
Vanhoutte PM. Other endothelium-derived vasoactive factors. Circulation 1993; 87(Suppl V): 9–17.
Moncada S, Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med 1993; 329: 2002–11.
Nozaki K, Moskowitz MA, Maynard KI, et al. Possible origins and distribution of immunoreactive nitric oxide synthase-containing nerve fibers in cerebral arteries. J Cereb Blood Flow Metab 1993; 13: 70–9.
Toda N. Mediation by nitric oxide of neurally-induced human cerebral artery relaxation. Experientia 1993; 49: 51–3.
Blaise GA, Sill JC, Nugent M, Van Dyke RA, Vanhoutte PM. Isoflurane causes endothelium-dependent inhibition of contractile responses of canine coronary arteries. Anesthesiology 1987; 67: 513–7.
Park KW, Dai HB, Lowenstein E, Darvish A, Sellke FW. Heterogeneous vasomotor responses of rabbit coronary microvessels to isoflurane. Anesthesiology 1994; 81: 1190–7.
Greenblatt EP, Loeb AL, Longnecker DE. Endotheliumdependent circulatory control — a mechanism for the differing peripheral vascular effects of isoflurane versus halothane. Anesthesiology 1992;77: 1178–85.
Uggeri MJ, Proctor GJ, Johns RA. Halothane, enflurane, and isoflurane attenuate both receptor-and non-receptor-mediated EDRF production in rat thoracic aorta. Anesthesiology 1992; 76: 1012–7.
Toda H, Nakamura K, Hatano T, Nishiwada M, Kakuyama M, Mori K. Halothane and isoflurane inhibit endothelium-dependent relaxation elicited by acetylcholine. Anesth Analg 1992; 75: 198–203.
Nakamura K, Temsako K, Toda H, et al. Mechanims of inhibition of endothelium-dependent relaxation by halothane, isoflurane, and sevoflurane. Can J Anaesth 1994; 41: 340–6.
Blaise G, To Q Parent M, Lagarde B, Asenjo F, Sauvé R. Does halothane interfere with the release, action, or stability of endothelium-derived relaxing factor/nitric oxide? Anesthesiology 1994; 80: 417–26.
Lerman J, Willis MM, Gregory GA, Eger EI II. Osmolarity determines the solubility of anesthetics in aqueous solutions at 37°C. Anesthesiology 1983; 59: 554–8.
Bollen BA, Tinker JH, Hermsmeyer K. Halothane relaxes previously constricted isolated porcine coronary artery segments more than isoflurane. Anesthesiology 1987; 66: 748–52.
Zuo Z, Johns RA. Halothane, enflurane, and isoflurane do not affect the basal or agonist-stimulated activity of partially isolated soluble and particulate guanylyl cyclases of rat brain. Anesthesiology 1995; 83: 395–404.
Stone JD, Johns RA. Endothelium-dependent effects of halothane, enflurane, and isoflurane on isolated rat aortic vascular rings. Anesthesiology 1989; 71: 126–32.
Hossain MD, Evers AS. Volatile anesthetic-induced efflux of calcium from IP3-gated stores in clonal (GH3) pituitary cells. Anesthesiology 1994; 80: 1379–89.
Iino M, Kobayashi T, Endo M. Use of ryanodine for functional removal of the calcium store in smooth muscle cells of the guinea-pig. Biochem Biophys Res Commun 1988; 152: 417–22.
Missiaen L, De Smedt H, Droogmans G, Himpens B, Casteels R. Calcium ion homeostasis in smooth muscle. Pharmacol Ther 1992; 56: 191–231.
Su JY, Bell JG. Intracellular mechanism of action of isoflurane and halothane on striated muscle of the rabbit. Anesth Analg 1986; 65: 457–62.
Su JY, Kerrick WGL. Effects of enflurane on functional skinned myocardial fibers from rabbits. Anesthesiology 1980; 52: 385–9.
Su JY, Chang YI, Tang LJ. Mechanisms of action of enflurane on vascular smooth muscle. Anesthesiology 1994; 81: 700–9.
Buljubasic N, Rusch NJ, Marijic J, Kampine JP, Bosnjak ZJ. Effects of halothane and isoflurane on calcium and potassium channel currents in canine coronary arterial cells. Anesthesiology 1992; 76: 990–8.
Eskinder H, Rusch NJ, Supan ED, Kampine JP, Boznjak ZJ. The effects of volatile anesthetics on L-and T-type calcium channel currents in canine cardiac Purkinje cells. Anesthesiology 1991; 74: 919–26.
Bolton TB. Mechanisms of action of transmitters and other substances on smooth muscle. Physiol Rev 1979; 59: 606–18.
Godfraind T, Dieu D. The inhibition by flunarizine of the norepinephrine-evoked contraction and calcium influx in rat aorta and mesenteric arteries. J Pharmacol Exp Ther 1981; 217: 510–5.
Akata T, Nakashima M, Kodama K, Boyle WA III, Takahashi S. Effects of volatile anesthetics on acetylcholine-induced relaxation in the rabbit mesenteric resistance artery. Anesthesiology 1995; 82: 188–204.
Boulanger C, Hendrickson H, Lorenz RR, Vahoutte PM. Release of different relaxing factors by cultured porcine endothelial cells. Circ Res 1989; 64: 1070–8.
Hoeffner U, Feletou M, Flavahan NA, Vanhoutte PM. Canine arteries release two different endothelium-derived relaxing factors. Am J Physiol 1989; 257: H330–3.
Hecker M, Bara AT, Bauersachs J, Busse R. Characterization of endothelium-derived hyperpolarizing factor as a cytochrome P450-derived arachidonic acid metabolite in mammals. J. Physiol (Lond) 1994; 481: 407–14.
Bauersachs J, Hecker M, Busse R. Display of the characteristics of endothelium-derived hyperpolarizing factor by a P450-derived arachidonic acid metabolite in the coronary microcirculation. Br J Pharmacol 1994; 113: 1548–53.
Fulton D, Mahboubi K, McGiff JC, Quilley J. Cytochrome P450-dependent effects of bradykinin in the rat heart. Br J Pharmacol 1995; 114: 99–102.
Bolotina VM, Najibi S, Palacino JJ, Pagano JP, Cohen RA. Nitric oxide directly activates calcium-dependent potassium channels in vascular smooth muscle. Nature 1994; 368: 850–3.
Johns RA, Tichotsky A, Muro M, Spaeth JP, Le Cras TD, Rengasamy A. Halothane and isoflurane inhibit endothelium-derived relaxing factor-dependent cyclic guanosine monophosphate accumulation in endothelial cell-vascular smooth muscle co-cultures independent of an effect on guanylyl cyclase activation. Anesthesiology 1995; 83: 823–34.
Blaise G, To Q Sauvé R. Superoxide dismutase (SOD) can suppress halothane interaction with nitric oxide (NO) stability. Anesthesiology 1994; 81: A770.
Author information
Authors and Affiliations
Additional information
This work was funded by the Medical Research Council of Canada.
Rights and permissions
About this article
Cite this article
Blaise, G., Guy, C., To, Q. et al. Do enflurane and isoflurane interfere with the release, action, or stability of endothelium-derived relaxing factors?. Can J Anaesth 44, 550–558 (1997). https://doi.org/10.1007/BF03011946
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03011946