Abstract
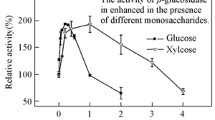
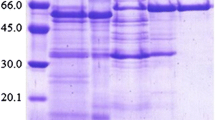
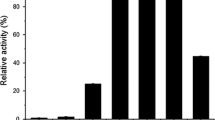
Purified β-glucosidase fromCellulomonas biazotea had an apparentK m andV for 2-nitrophenyl β-d-glucopyranoside (oNPG) of 0.416 mmol/L and 0.22 U/mg protein, respectively. The activation energy for the hydrolysis of pNPG of β-glucosidase was 65 kJ/mol. The inhibition by Mn2+ vs. oNPG of parental β-glucosidase was of mixed type with apparent inhibition constants of 0.19 and 0.60 µmol/L for the enzyme and enzyme-substrate complex, respectively. Ethanol at lower concentrations activated while at higher concentrations it inhibited the enzyme. The determination of apparent pK a’s at different temperatures and in the presence of 30 % dioxane indicated two carboxyl groups which control theV value. The thermal stability of β-glucosidase decreased in the presence of 10 % ethanol. The half-life of β-glucosidase in 1.75 mol/L urea at 35 °C was 145 min, as determined by 0–9 mol/L transverse urea gradient-PAGE.
Similar content being viewed by others
References
Barron A., May G., Berner E., Villarejo M.: Regulation of envelope protein composition during adaptation to osmotic stress inEscherichia coli.J.Bacteriol. 167, 433–438 (1986).
Clarke A.J., Bray M.R., Starting H.: β-Glucosidase, β-glucanases and xylanases: Their mechanism of catalysis, pp. 27–41 inβ-Glucosidase, Biochemistry and Molecular Biology (A. Esen, Ed.). American Chemical Society, Washington (DC) 1993.
Coughlan M.P.: Staining techniques for the detection of the individual components of cellulolytic enzyme system.Methods Enzymol. 160, 135–144 (1988).
Dixon M., Webb E.C.: Enzyme kinetics, pp. 47–206 inEnzymes. Academic Press, New York 1979a.
Dixon M., Webb E.C.: Enzyme inhibition and activation, pp. 332–467 inEnzymes. Academic Press, New York 1979b.
Esen A.: β-Glucosidases, over review, pp. 1–14 inβ-Glucosidase, Biochemistry and Molecular Biology (A. Esen, Ed.). American Chemical Society, Washington (DC) 1993.
Fersht A.:Enzyme Structure and Mechanism. W.H. Freeman & Co., New York 1985.
Goldenberg D.P.: Analysis of protein conformation by gel electrophoresis, pp. 225–250 inProtein Structure: A Practical Approach (T.E. Creighton, Ed.). IRL Press, Oxford (UK) 1989.
Heupel C., Schlochtermeier A., Schrempf H.: Characterization of an intracellular β-glucosidase fromStreptomyces reticuli.Enzyme Microb.Technol. 15, 127–132 (1993).
Kishimoto N., Inagaki K., Sugio T., Tano T.: Purification and properties of an acidic β-glucosidase fromAcidobacterium capsulatum.J.Ferment.Bioeng. 71, 318–321 (1991).
Nahrstedt A., Mueller E.: β-Glucosidase (linamarase) of the larvae of the mothZygaena trifolii and its inhibition by some alkaline earth metal ions, pp. 132–144 inβ-Glucosidase, Biochemistry and Molecular Biology (A. Esen, Ed.). American Chemical Society, Washington (DC) 1993.
Rajoka M.I., Malik K.A.: Comparison of different strains ofCellulomonas for production of cellulolytic and xylanolytic enzymes from biomass produced on saline lands.Biotechnol.Lett. 8, 557–560 (1986).
Rangarajan M., Hartley B.S.: Mechanism ofd-fructose isomerization byArthrobacter d-xylose isomerase.Biochem.J. 283, 223–233 (1992).
Sanyal A., Kundu R.K., Dube S., Dube D.K.: Extracellular cellulolytic enzyme system ofAspergillus japonicus. 2. Purification and characterization of an inducible extracellular β-glucosidase.Enzyme Microb.Technol. 10, 91–99 (1988).
Siddiqui K.S., Azhar M.J., Rashid M.H., Rajoka M.I.: Activity and thermostability of carboxymethylcellulase fromAspergillus niger is strongly influenced by noncovalently attached polysaccharides.World J.Microbiol.Biotechnol. 12, 213–216 (1996a).
Siddiqui K.S., Rashid M.H., Ghauri T.M., Durrani I.S., Rajoka M.I.: Purification and characterization of an intracellular β-glucosidase fromCellulomonas biazotea. World J.Microbiol.Biotechnol., in press (1996b).
Tipton K.F., Dixon H.B.F.: Effect of pH on enzymes.Methods Enzymol. 63, 183–234 (1979).
Trimbur D., Warren R.A.J., Withers S.G.: A β-glucosidase from anAgrobacterium sp., pp. 42–55 inβ-Glucosidase, Biochemistry and Molecular Biology (A. Esen, Ed.). American Chemical Society, Washington (DC) 1993.
Tsitsimpikou C., Voutou D., Christakopoulos P., Kekos D., Macris B.J., Kolisis F.N.: Studies of the effect of organic solvents on the stability of β-glucosidase fromFusarium axysporum.Biotechnol.Lett. 16, 57–62 (1994).
Vaserot Y., Christiaens H., Chemardin P., Arnauod A., Galzy P.: Purification and properties of a β-glucosidase ofHanseniospora vineae Van der Walt andTscheuschner with the view to its utilization in fruit aroma liberation.J.Appl.Bacteriol. 66, 271–279 (1989).
Wood T.M., Bhat K.M.: Methods for measuring cellulase activities.Methods Enzymol. 160, 87–112 (1988).
Author information
Authors and Affiliations
Additional information
This work was financed in part by a grant made by theUS Agency for International Development under PSTC proposal 6-163,USAID grant no. 9365542-G-00-89-42-00, and PAEC.
Rights and permissions
About this article
Cite this article
Siddiqui, K.S., Rashid, M.H. & Rajoka, M.I. Kinetic analysis of the active site of an intracellular β-glucosidase fromCellulomonas biazotea . Folia Microbiol 42, 53–58 (1997). https://doi.org/10.1007/BF02898646
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02898646