Abstract
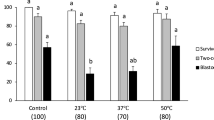
Conditions were evaluated for optimum cryopreservation of primary chicken embryo kidney (CEK) cells. The recovery of viable CEK cells was best (50.8% viability) when the concentration of dimethyl sulfoxide (DMSO) in the freezing medium was 20% (v/v). The viability of primary CEK cells was not influenced by the concentration of calf serum in the freezing medium, the duration of storage at −70°C before storage in liquid nitrogen, cell concentration, or the method of addition or dilution of DMSO.
Thawed cells recovered and grew in complete growth medium similarly to cells freshly isolated from kidney, and influenza viruses produced plaques in the monolayer. The cryopreservation procedures described here may facilitate maintenance of a standard stock of primary CEK cells for laboratories where preparation of primary CEK cells is not an option.
Similar content being viewed by others
References
Freshney, R. I. (2000) Culture of Animal Cells: A Manual of Basic Technique, 4th ed. Wiley-Liss Inc., New York.
Hay, A. J., Belshe, R. B., Anderson, E. L., Gorse, G. J., and Westblom, T. U. (1991) Textbook of Human Virology, 2nd ed., Mosby Year-Book Inc., St. Louis, MO. pp. 307–371.
Subbarao, K., Klimov, A., Katz, J., et al. (1998) Characterization of an avian influenza A (H5N1) virus isolated from a child with a fatal respiratory illness. Science 279, 393–396.
Maassab, H. F. (1969) Biologic and immunologic characteristics of cold-adapted influenza virus. J. Immunol. 102, 728–732.
Tannock, G. A., Bryce, D. A., and Paul, J. A. (1985) Evaluation of chicken kidney and chicken embryo kidney cultures for the large-scale growth of attenuated influenza virus master strain A/Ann/Arbor/6/60-ca. Vaccine 3, 333–339.
Chagnon, A., and Pavilanis, V. (1966) Freezing and storing of monkey and rabbit kidney cells for tissue cultures. Cryobiol. 3, 81–84.
Chagnon, A., Couture, B., and Pavilanis, V. (1968) Some optimal conditions in freezing rabbit kidney cells for tissue cultures. Cryobiol. 4, 197–199.
Lindsey, N. J. and Chow, T. L. (1969) Preservation of primary bovine embryonic kidney cells with dimethyl sulfoxide. Appl. Microbiol. 17, 484–485.
Wolfe, J. and Bryant, G. (2001) Cellular cryobiology: thermodynamic and mechanical effects. Int. J. Refrigeration 24, 438–450.
Stephens, E. A. and Sharma, J. M. (1981) Cryopreservation of avian lymphoid cells. Avian Dis. 25, 614–627.
Gao, D. and Crister, J. K. (2004) Mechanisms of cryoinjury in living cells. ILAR J. 41, 187–196.
Ashwood-Smith, M. J. (1967) Radioprotective and cryoprotective properties of dimethyl sulfoxide in cellular system. Ann. NY Acad. Sci. 141, 45–62.
Price, P. J., Anderson, B. P., and Bell, M. J. (1966) Uncontrolled and controlled rates of freezing of human kidney cell suspensions. Cryobiol. 3, 269–271.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, JW., Shin, EK., Ha, SH. et al. Optimal conditions for cryopreservation of primary chicken embryo kidney cells with dimethyl sulfoxide. Mol Biotechnol 35, 237–241 (2007). https://doi.org/10.1007/BF02686009
Issue Date:
DOI: https://doi.org/10.1007/BF02686009