Summary
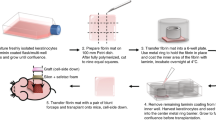
The attachment, proliferation, and differentiation of primary cultures of kerationcytes isolated from murine epidermis were monitored after purified cell suspensions were seeded and incubated in vitro on various synthetic membranes. Concomitant studies of the effects of attachment factors added to synthetic membranes before use as substrata for keratinocytes were also done. The study demonstrated that a synthetic membrane composed of nylon was superior to other membranes and to plastic control culture vessels in supporting the growth of murine keratinocytes. Although laminin enhanced initial attachment and proliferation of cells on nylon membranes, the untreated substratum was more effective for extended incubation. Stratification and differentiation of these kerationcytes on the nylon substratum was enhanced by raising confluent cultures (7 d) to the air-medium interface so that they were in contact with medium only from the bottom. Cultures raised for 14 d produced many morphologic markers of the epidermis and closely resembled the architecture of this tissue in situ.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Carlsson, R.; Engvall, E.; Freeman, A., et al. Laminin and fibronectin in cell adhesion: Enhanced adhesion of cells from regenerating liver to laminin. Proc. Natl. Acad. Sci. USA 78:2403–2406; 1981.
Cornell, P. The use of nuclepore filters in ultrastructural studies of cell cultures. Exp. Cell Res. 56:156–158; 1969.
England, M. A. Millipore filters studies in isolation and in vitro by transmission electron microscopy and stereoscaning electron microscopy. Exp. Cell. Res. 54:222–230; 1969.
Flaxman, B. A. Cell identification in primary cell cultures from skin. In Vitro 10:112–118; 1974.
Freeman, A. E.; Igel, U. J.; Herrman, B. J., et al. Growth characterization of human skin epithelial cell cultures. In Vitro 12:352–362; 1976.
Fusenig, N. E.; Amer, S. M.; Boukamp, P., et al. Characteristics of chemically transformed mouse epidermal cells in vitro and in vivo. Bull. Cancer 65:271–280; 1978.
Gilchrest, B. A.; Calhoun, J. K.; Maciag, T. Attachment and growth of human keratinocytes in a serum-free environment. J. Cell. Physiol. 112:197–206; 1982.
Green, H. Terminal differentiation of cultured human epidermal cells. Cell 11:405–416; 1977.
Grobstein, C. Mechanisms of organogenetic tissue interaction. Natl. Cancer Inst. Monogr. 26:279; 1976.
Ham, R. G.; McKeehan, W. L. Nutritional requirements for clonal growth of nontransformed cells. In: Katsuta, H., ed. Nutritional requirements of cultured cells. Tokyo: Japan Scientific Societies Press; 1978:63–155.
Hennings, H.; Michael, D.; Cheng, C., et al. Calcium regulation of growth and differentiation of mouse epidermal cells in culture. Cell 19:245–254; 1980.
Holbrook, K. A.; Hennings, H. Phenotypic expression of epidermal cells in vitro: A review. J. Invest. Dermatol. 81 (1 Suppl): 11s-24s; 1983.
Karasek, M. A.; Charlton, M. E. Growth of postembryonic skin epithelial cells on collagen gels. J. Invest. Dermatol. 56:205–210; 1971.
Lillie, J. H.; MacCallum, D. K.; Jepsen, A. Fine structure of subcultivated stratified squamous epithelium grown on collagen rafts. Exp. Cell Res. 125:153–165; 1980.
Liu, S. C.; Karasek, M. A. Isolation and growth of adult human epidermal keratinocytes in cell culture. J. Invest. Dermatol. 71:157–162; 1978.
Maciag, T.; Nemore, R. E.; Weinstein, R., et al. An endocrine approach to the control of epidermal growth: Serum-free cultivation of human keratinocytes. Science 211:1452–1454; 1981.
Michalopoulos, G.; Pitot, H. C. Primary culture of parenchymal liver cells on collagen membranes. Exp. Cell Res. 14:70–78; 1975.
Murray, J. C.; Stingl, G.; Kleinman, H. K., et al. Epidermal cells adhere preferentially to type IV (basement membrane) collagen. J. Cell Biol. 80:197–202; 1979.
Okada, N.; Kitano, Y.; Ichihara, K. Effects of cholera toxin on cultured human keratinocytes in relation to intracellular cyclic AMP levels. J. Invest. Dermatol. 79:42–47; 1982.
Peehl, D. M.; Ham, R. G. Clonal growth of human keratinocytes with small amounts of dialyzed serum. In Vitro 16:526–450; 1980.
Price, F. M.; Camalier, R. F.; Gantt, R., et al. A new culture medium for human skin epithelial cells. In Vitro 16:147–158; 1980.
Prunieras, M. D.; Delescluse, D.; Regnier, M. J. Growth and differentiation of postembryonic mammalian epidermal keratinocytes in culture. Front. Matrix. Biol. 3:52–76; 1976.
Prunieras, M.; Regnier, M.; Woodley, D. Methods for cultivation of keratinocytes with an air-liquid interface. J. Invest. Dermatol. 81 (1 Suppl): 28s-33s; 1983.
Regnier, M.; Prunieras, M.; Woodley, D. Growth and differentiation of adult human epidermal cells on dermal substrates. Front. Matrix. Biol. 9:4–35; 1981.
Rheinwald, J. G.; Green, H. Formation of a keratinizing epithelium in culture by a cloned cell line derived from a teratoma. Cell 6:317–330; 1975.
Tsao, M. C.; Walthall, B. J.; Ham, R. C. Clonal growth of normal human epidermal keratinocytes in a defined medium. J. Cell. Physiol. 110:219–229; 1982.
Terranova, V. P.; Rohrbach, D. H.; Martin, G. R. Role of laminin in the attachment of PAM 212 (epithelial) cells to basement membrane collage, Cell 22:719–726; 1980.
Vaughan, F. L.; Bernstein, I. A. Studies of proliferative capabilities in isolated epidermal basal and differentiated cells. J. Invest. Dermatol. 56:454–466; 1971.
Vaughan, F. L.; Bernstein, I. A.; Freed, P., et al. The use of biological processes in the development of a long-term percutaneous energy transmission system. In: Szycher, M., ed. Biocompatible polymers, metals and ceramics. Lancaster: Technomic Publishing Co.; 1983:621–655.
Vaughan, F. L.; Kass, L. L.; Uzman, J. A. Requirement of hydrocortisone and insulin for extended proliferation and passage of rat keratinocytes. In Vitro 17:941–946; 1981.
Waymouth, C. Studies on chemically defined media and the nutritional requirements of cultures of epithelial cells. In: Katsuta, H., ed. Nutritional requirements of cultured cells. Tokyo: Japan Scientific Societies Press; 1978:38–61.
Author information
Authors and Affiliations
Additional information
This investigation was supported in part by the Johns Hopkins Center for Alternatives to Animal Testing and by the United States Army Medical Research and Development command, contract DAMD17-82-C-2198. The views, opinions or findings contained in this report are those of the authors and should not be construed as position, policy or decision of the agency supporting the research.
Rights and permissions
About this article
Cite this article
Vaughan, F.L., Gray, R.H. & Bernstein, I.A. Growth and differentiation of primary rat kerationocytes on synthetic membranes. In Vitro Cell Dev Biol 22, 141–149 (1986). https://doi.org/10.1007/BF02623501
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02623501