Abstract
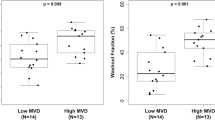
Since detailed knowledge regarding the pathophysiological properties—which in turn are responsible for differences in contrast enhancement—remain fairly undetermined, it was the aim of this study (i) to examine the association of standard and pharmacokinetic analysis of time-intensity curves in dynamic MRI with histomorphological markers of tumor angiogenesis (microvessel density [MVD]; vascular endothelial growth factor [VEGF]) and (ii) to determine the ultimate value of a histomorphological and a dynamic MRI approach by correlation of those data with disease outcome in patients with primary cancer of the uterine cervix. Pharmacokinetic parameters (amplitude A, exchange rate constantk 21) and standard parameters (maximum signal intensity (SI)-increase, [SI-I] over baseline and steepest SI-upslope, per second [SI-U/s]) were calculated from contrast-enhanced dynamic MR imaging series in 37 patients with biopsy-proven primary cervical cancer. On the surgical whole mount specimens, histomorphological markers of tumor angiogenesis (MVD, VEGF) were compared with similar sized and positioned regions-of-interest (ROIs) on the MRI-derived parameters. For MRI and histomorphological data, Kaplan-Meier survival curves were calculated and compared using log-rank statistics. A significant association was found betweenMVD and amplitudeA (P<0.01) andSI-I (P<0.05). No significant relationships were observed between theVEGF expression and all dynamic MRI parameters. Kaplan-Meier curves based onk 21 and SI-U/s showed that tumors with highk 21 andSI-U/s values had a significantly (P<0.05, 0.001, respectively) worse disease outcome than tumors with lowk 21 andSI-U/s values. None of the histomorphological gold standard markers for assessing tumor angiogenesis (MVD, VEGF) had any significant power to predict patient survival. It is concluded that (1) the pathophysiological basis for differences in dynamic MRI isMVD but not VEGF-expression; (2) a functional, dynamic MRI approach (both standard and a pharmacokinetic analysis) may be better suited to assess angiogenic activity in terms of patient survival than current histomorphological-based markers of tumor angiogenesis; and [3] compared with standard analysis, a simple pharmacokinetic analysis of time-intensity curves is not superior to assessMVD or patient survival.
Similar content being viewed by others
References
Folkman J. Intratumoral microvascular density as a prognostic factor in cancer. Am J Pathol 1995;147:9–19.
Weidner N, Joseph PS, William RW, Folkman J. Tumor angiogenesis and metastases-correlation in invasive breast carcinoma. N Engl J Med 1991;324:1–8.
Macciarini P, Fontanini G, Hardin MJ, Squartini F, Angeletti CA. Relation of neovascularization to metastasis of non-small-cell lung cancer. Lancet 1992;340:145–6.
Wiggins DL, Granai CO, Steinhoff MM, Calabresi P. Tumor angiogenesis as a prognostic factor in cervical carcinoma. Gynecol Oncol 1995;56:353–6.
Schlenger K, Hoeckel M, Mitze M, Schaeffer U, Weikel W, Knapstein PG, Lambert A. Tumor vascularity-a novel prognostic factor in advanced cervical carcinoma. Gynecol Oncol 1995;59:57–66.
Bernsen HJJA, Rijken PFJW, Oostendorp T, van der Kogel AJ. Vascularity and perfusion of human gliomas xenografted in the athymic nude mouse. Br J Cancer 1995;71:721–6.
Brown LF, Berse B, Jackman RW, Tognazzi K, Guidi AJ, Dvorak HF, et al. Expression of vascular permeability endothelial growth (factor) and its receptors in breast cancer. Human Pathol 1995;26:86–91.
Kim KJ, Li B, Winer J, Armanini M, Gillett N, Phillips HS. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumor growth in vivo. Nature 1993;362:841–4.
Verstraete KL, De-Deene YD, Roels H, Dierick A, Uyttendaele D, Kunnen M. Benign and malignant musculoskeletal lesions: dynamic contrast-enhanced MR imaging: parametric first-pass images depict tissue vascularization and perfusion. Radiology 1994;192:835–43.
Buadu LD, Murakami J, Murayama S, Hashiguchi N, Sakai S, Masuda K, et al. Breast lesions: correlation of contrast medium enhancement patterns on MR images with histopathologic findings and tumor angiogenesis. Radiology 1996;200:639–49.
Degani H, Gussi V, Weinstein D, Fields S, Strano S. Mapping pathophysiological features of breast tumors by MRI at high spatial resolution. Nat Med 1997;3:780–2.
Hoffmann U, Brix G, Knopp MV, Hess T, Lorenz WJ. Pharmacokinetic mapping of the breast: a new method for dynamic MR-mammography. Magn Reson Med 1995;33:506–14.
Brix G, Semmler W, Port R, Schad LR, Layer G, Lorenz WJ. Pharmacokinetic—parameters in CNS Gd-DTPA enhanced MR imaging. J Comput Assist Tomogr 1991;15:621–727.
Koenig H, Sieper J, Wolf KJ. Rheumatoid arthritis: evaluation of hypervascular and fibrous pannus with enhanced dynamic MR imaging enhanced with Gd-DTPA. Radiology 1990;176:473–7.
Hawighorst H, Knapstein PG, Weikel W, Knopp MV, Schaeffer U, Brix G, et al. Cervical carcinoma: comparison of standard and pharmacokinetic MR imaging. Radiology 1996;201:531–9.
Hawighorst H, Knapstein PG, Schaeffer U, Knopp MV, Brix G, Hoffmann U, et al. Pelvic lesions in patients with treated cervical-carcinoma: efficacy of pharmacokinetic MR images in distinguishing recurrent tumors from benign conditions. Am J Radiol 1996;166:4012–8.
Hawighorst H, Weikel W, Knapstein PG, Knopp MV, Zuna I, Schonberg SO, et al. Clin Cancer Res (in press).
Takashima S, Noguchi Y, Okumara T, Aruga H, Kobayashi T. Dynamic MR imaging in the head and neck. Radiology 1993;189:813–21.
Hawighorst H, Knapstein PG, Weikel W, Knopp MV, Zuna I, et al. Angiogenesis of uterine cervical carcinoma: characterization by pharmacokinetic magnetic resonance parameters and histological microvessel density with correlation to lymphatic involvement. Cancer Res 1997;57:4777–86.
Mattern J, Koomagi R, Volm M. Association of vascular endothelial growth-factor expression with intratumoral microvessel density and tumor cell proliferation in human epidermoid carcinoma. Br J Cancer 1996;73:931–4.
Neeman M, Abramovitch R, Schiffenbauer YS, Tempel C. Regulation of angiogenesis by hypoxic stress: from solid tumors to the ovarian follicle. Int J Exp Path 1997;78:57–70.
Shweiki D, Neeman M, Itin A, Keshet E. Induction of vascular endothelial growth factor expression by hypoxia and by glucose deficiency in multiple spheroids: implications for tumor angiogenesis. Proc Natl Acad Sci USA 1995;92:768–72.
Stein I, Neeman M, Shweiki D, Itin A, Keshet E. Stabilization of vascular-endothelial growth factor mRNA by hypoxia and hypoglycemia and coregulation with other ischemia-induced genes. Mol Cell Biol 1995;15:5363–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hawighorst, H., Knapstein, P.G., Knopp, M.V. et al. Cervical carcinoma: standard and pharmacokinetic analysis of time-intensity curves for assessment of tumor angiogenesis and patient survival. MAGMA 8, 55–62 (1999). https://doi.org/10.1007/BF02590636
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02590636