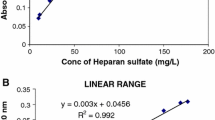
Abstract
Glycosaminoglycans can stimulate the synthesis of glomerular basement membrane (GBM) protein by glomerular cells and correct biochemical alterations of the GBM. In this study we examine the effects of heparin and low-molecular-weight heparin (LMWH) on glomerular permeability to proteins and glomerular structure in Adriamycin-treated rats. One Adriamycin dose of 5 mg/kg body weight was administered i.v. to 38 female Wistar rats. Eleven animals were also treated with heparin (250 U) administered s.c. twice daily and 7 with LMWH 6 mg/kg body weight administered s.c. twice daily. Urine samples were collected before and 30 and 60 days after the beginning of treatment to quantify albumin excretion and to characterize urinary proteins by gel filtration and electrophoresis in sodium dodecyl sulfate polyacrylamide gels. Blood samples were also collected on day 60 from these rats to estimate renal permeability by gel filtration; the rats were then killed and the kidneys removed for histological analysis. Heparin administration caused a reduction in urinary albumin excretion and in the incidence of segmental glomerulosclerosis in Adriamycin-treated rats. However, heparin did not modify the selectivity of the GBM to proteins of different molecular weights. These data suggest that the effect of heparin on albuminuria may be due to changes in the negative GBM charges induced by this drug.
Similar content being viewed by others
References
Bertani T, Poggi A, Pozzoni R, DeLaini F, Sacchi G, Thoua Y, Mecca G, Remuzzi G, Donati MB (1982) Adriamycin-induced nephrotic syndrome in rats. Lab Invest 46:16–23
Weening JJ, Rennke HG (1983) Glomerular permeability and polianion in adriamycin nephrosis in the rat. Lab Invest 24:152–159
Bertolatus JA, Hunsiker LG (1985) Glomerular sieving of anionic and neutral bovine albumins in proteinuric rats. Kidney Int 28:467–476
Bertani T, Cutillo F, Zoja C, Brogginne M, Remuzzi G (1986) Tubulo-interstitial lesions mediate renal damage in adriamycin glomerulopathy. Kidney Int 30:488–496
Bertani T, Rocchi G, Sacchi G, Mecca G, Remuzzi G (1986) Adriamycin-induced glomerulosclerosis in the rat. Am J Kidney Dis 7:12–19
Okuda S, Oh Y, Tsuruda H, Onoyama K, Fujimi S, Fujishima M (1986) Adriamycin-induced nephropathy as a model of chronic progressive glomerular disease. Kidney Int 29:502–510
Gambaro G, Carvazzana AO, Luzi P, Piccoli A, Borsatti A, Crepaldi G, Marchi E, Venturini AP, Baggio B (1992) Glycosaminoglycans prevent morphological renal alterations and albuminuria in diabetic rats. Kidney Int 42:285–291
Gambaro G, Venturini AP, Noonan DM, Fries W, Garbisa GS, Milanesi C, Pesarini A, Borsatti A, Marchi E, Baggio B (1994) Treatment with glycosaminoglycan formulation ameliorates experimental diabetic nephropathy. Kidney Int 46:797–806
Diamond JR, Karnovsky MJ (1986) Nonanticoagulant protective effect of heparin in chronic aminonucleoside nephrosis. Renal Physiol Biochem 9:366–374
Purkerson ML, Tolelfsen DM, Klahr S (1988) N-Desulfated-acetylated heparin ameliorates the progression of renal disease in rats with subtotal renal ablation. J Clin Invest 81:69–74
Ichikawa I, Yoshida Y, Fogo A, Purkerson ML, Klahr S (1988) Effect of heparin on the glomerular structure and function of remnant nephrons. Kidney Int 34:638–644
Laurell CB (1972) Electroimmunoassay. Scand J Clin Lab Invest 124 [Suppl 23]:21–23
Coimbra TM, Furtado MR, Lachat JJ, Carvalho IF (1983) Effects of administration of cationic and native homologous albumin on the kidney. Nephron 33:208–215
Hardwicke J (1965) Estimation of renal permeability to protein on Sephadex G 200. Clin Chim Acta 12:89–96
Granath KA, Kvist BE (1967) Molecular weight distribution analysis by gel chromatography on Sephadex. J Chromatogr 28:69–81
Laemmli UK (1970) Cleavage of structural proteins during the assembly of head bacteriophage T4. Nature 227:680–685
Hartree EF (1972) Determination of protein. A modification of Lowry method that gives a linear photometric response. Anal Biochem 48:422–427
Noël L-H, Zanetti M, Droz D, Barbanel B (1979) Long-term prognosis of idiopathic membranous glomerulonephritis. Am J Med 66:82–90
Hays WL (1973) Statistics for social sciences. Holt, Rinehart and Winston. New York, pp 605–607
Klein DJ, Oegema TR, Brown DM (1989) Release of glomerular heparan35SO4 proteoglycan by heparin from glomeruli of streptozotocin-induced diabetic rats. Diabetes 38:130–139
Nader HB, Buonassisi V, Colburn P, Dietrich CP (1989) Heparin stimulates the synthesis and modifies the sulfation pattern of heparan sulfate proteglycans from endothelial cells. J Cell Physiol 140:305–310
Olson JL (1984) Role of heparin as protective agent following reduction of renal mass. Kidney Int 25:376–382
Floege J, Eng E, Young BA, Couser WG, Johnson RJ (1993) Heparin suppresses mesangial cell proliferation and matrix expansion in experimental mesangioproliferative glomerulonephritis. Kidney Int 43:369–380
Castellot JJ, Hoover RL, Harper PA, Karnowsky MJ (1985) Heparin and glomerular epithelial cell-secreted heparin-like species inhibit mesangial cell proliferation. Am J Pathol 120:427–435
Person JM, Lovett DH, Raugi GJ (1988) Modulation of mesangial cell migration by extracellular matrix components. Inhibition by heparin-like glycosaminoglycans. Am J Pathol 133:609–614
Groggel GH, Marinides GN, Hovingh P, Hammond E, Linker A (1990) Inhibition of rat mesangial cell growth by heparan sulfate. Am J Physiol 258:F259-F265
Johnson RJ, Floege J, Couser WG, Alpers CE (1992) Role of platelet-derived growth factor in glomerular disease. J Am Soc Nephrol 4:119–128
Isaka Y, Fujiwara Y, Ueda N, Kaneda Y, Kamada T, Imai E (1993) Glomerulosclerosis induced by in vivo transfection of transforming growth factor B or platelet-derived growth factor gene into the kidney. J Clin Invest 92:2597–2601
Fogo A (1994) Internephron heterogenity of growth factors and sclerosis. Kidney Int [Suppl]45:S24-S26
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pieroni, F., Costa, R.S. & Coimbra, T.M. Effects of glycosaminoglycans on the glomerular changes induced by Adriamycin. Res. Exp. Med. 195, 373–379 (1995). https://doi.org/10.1007/BF02576808
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02576808