Abstract
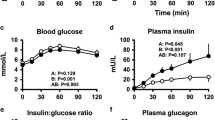
Systemic and regional haemodynamics and hormonal responses to two isovolaemic, iso-osmotic solutions of 0.5 and 1.0 g/kg of glucose were compared in ten normal young subjects (mean age 24±3 years). Measurements were made while subjects were supine before glucose and every 15 min for 60 min after ingestion of each solution. Superior mesenteric artery (SMA) blood flow rose similarly after each dose. There were corresponding reductions in SMA vascular resistance after each dose but no difference between doses. Pulsatility index of the SMA was lower after 1.0 g/kg. There was no change in blood pressure, heart rate, cardiac index or forearm muscle blood flow after either dose. Plasma glucose levels rose after each dose and were higher after 1.0 g/kg. There was no difference in the plasma insulin rise between doses. Plasma levels of noradrenaline and adrenaline did not change after either dose. These results suggest that with glucose loads within the ranges we used, changes in SMA blood flow are not dose-related and larger increases in SMA blood flow or in plasma insulin than we observed are needed to reflexly activate cardiac output and raise plasma noradrenaline levels in young normal subjects.
Similar content being viewed by others
References
Qamar MI, Read AE, Mountford R. Increased superior mesenteric blood flow after glucose but not lactulose ingestion.Quart J Med 1986;233: 893–896.
Sidery MB, Macdonald IA. The effect of meal size on the cardiovascular responses to meal ingestion.Brit J Nutr 1994;38: 234–242.
Mathias CJ, da Costa DF, Macintosh CM et al. Differential blood pressure and hormonal effects after glucose and xylose ingestion in chronic autonomic failure.Clin Sci 1989;77: 85–92.
Puvi-Rajasingham S, Kimber J, Akinola A, Magnifico F, Mathias CJ. Differences in regional haemodynamic responses to two oral glucose loads in humans with sympathetic denervation.J Physiol 1996;494P: 96P.
Huntsman L, Stewart D, Barnes S, Franklin S, Colocousis J, Hessel E. Non-invasive Doppler determination of cardiac output in man.Circulation 1983;67: 593–602.
Whitney R. The measurement of volume changes in human limbs.J Physiol 1953;121: 1–27.
Chaudhuri KR, Thomaides T, Hernandez P, Mathias CJ. Noninvasive quantification of superior mesenteric artery blood flow during sympathoneural activation in normal subjects.Clin Aut Res 1991;1: 37–42.
Qamar MI, Read AE, Skidmore R, Evans JM, Wells PNT. Pulsatility index of superior mesenteric artery blood velocity waveforms.Ultrasound in Medicine and Biology 1986;12: 773–776.
Gosling RG, King DH. Arterial assessment by Doppler shift ultrasound.Proc Roy Soc Med 1974;67(b): 447.
Skidmore R, Woodcock JP, Wells PNT, Bird D, Baird RND. Physiological interpretation of Doppler shift waveforms-III Clinical results.Ultrasound in Medicine and Biology 1980;6: 227–231.
Bloom SR, Long RG.Radioimmunoassay of Gut Regulatory Peptides. London: WB Saunders, 1982.
May C, Ham I, Heslop K, Stone F, Mathias CJ. Intravenous morphine causes hypotension, hypoglycaemia and increases sympathoadrenal activity in the conscious rabbit.Clin Sci 1991;75: 71–77.
Sidery MB, Macdonald IA, Cowley AJ, Fulwood LJ. Cardiovascular responses to high fat and high carbohydrate meals in young subjects.Am J Physiol 1991;261: H1430–1436.
Brown CM, Rees WDW, Gastroduodenal motility in health and disease. In: Bouchier IAD, Allan RN, Hodgson HJF, Keighley MRB, eds.Gastroenterology — Clinical Science and Practise. London: WB Saunders, 1993; 208–221.
Mathias CJ, Bannister R. Postcibal hypotension in autonomic disorders. In: Bannister R, Mathias CJ, eds.Autonomic Failure: A Textook of Clinical Disorders of the Autonomic Nervous System, 3rd edn. Oxford: Oxford University Press, 1993; 489–508.
Sit SP, Nyhof P, Gallavan R, Chou CC. Mechanisms of glucose-induced hyperaemia in the jejunum.Proc Soc Exp Biol Med 1980;163: 273–277.
Chaudhuri KR, Ryder SA, Thomaides T, Mathias CJ. The relationship between blood flow and pulsatility index measurement of the superior mesenteric artery at rest and during constrictor stimuli in normal subjects.J Clin Ultrasound 1994;22: 149–160.
Young JB, Rowe JW, Palotta JA, Sparrow D, Landsberg L. Enhanced plasma norepinephrine response to upright posture and oral glucose administration in elderly human subjects.Metabolism 1980;29(6): 532–539.
Hegedus L, Christensen NJ, Sestoft L. Abnormal regulation of sympathetic nervous system activity and heart rate after oral glucose in Type 1 (insulin-dependent) diabetic patients.Diabetologica 1983;25: 242–246.
Kooner JS, Peart WS, Mathias CJ. The peptide release inhibitor, Octreotide (SMS 201-995) prevents the hemodynamic change following food ingestion in normal human subjects.Quart J Exp Physiol 1989;74: 569–572.
Scott AR, Bennett T, Macdonald IA. Effects of hyperinsulinaemia on the cardiovascular responses to graded hypovolaemia in normal and diabetic subjects.Clin Sci 1988;75: 85–92.
Anderson EA, Hoffman RP, Balcon TW, Sinkey CA, Mark AL. Hyperinsulinaemia produces both sympathetic neural activation and vasodilation in normal humans.J Clin Invest 1991;87: 2246–2252.
Baron AD, Brechtel G. Insulin differentially regulates systemic and skeletal muscle vascular resistance.Am J Physiol 1993;265: E61-E67.
Kearney MT, Stubbs TA, Evans AE, Cowley AJ, Macdonald IA. Insulin mediated skeletal muscle vasodilation: a mechanism for postprandial hypotension.Clin Aut Res 1996;6(1): 51.
Christensen NJ, Hegedus L, Dejgaard A, Hilsted J. The cardiovascular effects of insulin. In: Christensen N, Henriksen O, Lassen N, eds.The Sympathoadrenal System. Alfred Benzon Symposium 23. Copenhagen: Munksgaard, 1986.
Welle S, Lilavivat U, Campbell RG. Thermic effect of feeding in man: increased plasma norepinephrine concentrations following gluose but not protein or fat consumption.Metabolism 1981;30: 953–958.
Welle S, Lilavivathana U, Campbell RG. Increased plasma norepinephrine concentrations and metabolic rates following glucose ingestion in man.Metab Clin Exp 1980;29: 806–808.
Santiago JV, Clarke WL, Shah SD, Cryer PE. Epinephrine, norepinephrine, glucagon and growth hormone release in association with physiological decrements in the plasma glucose concentration in normal and diabetic man.J Clin Endocr Metab 1980;51: 877–883.
Levine SE, Granger DN, Brace RA, Taylor AE. Effect of hyperosmolarity on vascular resistance and lymph flow in the cat ileum.Am J Physiol 1978;234: H14-H20.
Berne C, Fagius J, Niklasson F. Sympathetic response to oral carbohydrate administration: evidence from microelectrode recordings.J Clin Invest 1989;84: 1403–1409.
Tse T, Clutter WE, Shah SD, Miller JP, Cryer PE. Neuroendocrine responses to glucose ingestion in man.J Clin Invest 1983;72: 270–277.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Puvi-Rajasingham, S., Wijeyekoon, B., Natarajan, P. et al. Haemodynamic and hormonal effects of two different oral glucose loads in normal human subjects. Clinical Autonomic Research 7, 155–161 (1997). https://doi.org/10.1007/BF02308844
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02308844