Abstract
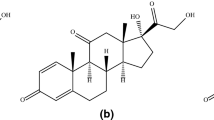
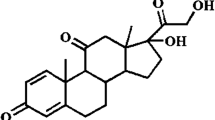
The kinetics of decomposition of prednisolone in aqueous solution were studied under anaerobic conditions at 100°C within the pH range 1.8–8.3. At pH=2.5 prednisolone is most stable. Between pH=5.0 and pH=6.0 the decomposition rate is nearly independent of the pH value. The decomposition products, 17-deoxy-21-dehydroprednisolone, 17-deoxyprednisolone, the 17-ketosteroid and the D-homosteroid were analysed quantitatively. The total amount of acidic decomposition products was approximated. Below pH=5.5 prednisolone decomposes to 17-deoxy-21-dehydroprednisolone. However, at higher pH values a mixture of decomposition products appears. All analyses were performed by a stability indicating high performance liquid Chromatographic method using uv detection.
Similar content being viewed by others
References
Bundgaard, H., andJ. Hansen (1979)Arch. Pharm. Chemi Sci. Ed. 7, 19.
Conbere, J. P., and N. J.Fanwood (1956)US patent 2, 773,077.
Dekker, D. (1979)Pharm. Weekblad Sci. Ed. 1, 112;Ibidem (1980a)2, 14;Ibidem (1980b)2, 28;Ibidem (1980c)2, 59;Ibidem (1980d)2, 87.
Dekker, D., andJ. H. Beijnen (1980a)J. Chromtog. 193, 480;Ibidem (1980b)Fresenius Z. Anal. Chem. in press.
Dekker, D., andD. J. Buijs (1980)Pharm. Weekblad Sci. Ed. 2, 54.
Duvivier, J. (1965)J. Chromatog. 19, 352.
Guttman, D. E., andP. D. Meister (1958)J. Am. Pharm. Assoc. Sci. Ed. 47, 773.
Hansen, J., andH. Bundgaard (1979)Arch. Pharm. Chemi Sci. Ed. 7, 135.
Kripalani, K. J., andD. L. Sorby (1967)J. Pharm. Sci. 56, 687.
Mason, H. L. (1938)J. Biol. Chem. 124, 475.
Monder, C. (1968)Endocrinology 82, 318.
Oesterling, T. O., andD. E. Guttman (1964)J. Pharm. Sci. 53, 1189.
Wendler, N. L., and R. P.Graber (1956)Chem. and Ind. (London), 549.
Author information
Authors and Affiliations
Additional information
Part v, seeDekker, D. (1980)Pharm. Weekblad Sci. Ed. 2, 87.
Rights and permissions
About this article
Cite this article
Dekker, D., Beijnen, J.H. Stability of corticosteroids under anaerobic conditions. Pharmaceutisch Weekblad Scientific Edition 2, 1116–1120 (1980). https://doi.org/10.1007/BF02273268
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02273268