Abstract
Through a linker containing thiocarbomate bound to the 7-N position of mitomycin C (MMC), conjugates with a monoclonal antibody to CD10 (NL-1) were prepared, and their antitumor activities were examined. All five conjugates, except one, showedin vitro cytotoxity to two CD10+ lymphoid cell lines superior to MMC. The conjugate displaying the highest cytotoxicity was selected and further tested against three CD10+ and two CD10 lymphoid cell linesin vitro. The conjugate with NL-1 antibody demonstrated higher cytotoxic activity against CD10+ tumor cells than the control conjugate with normal immunoglobulin, while there was no significant difference, when tested against CD10− tumors. The cytotoxic activity of the NL-1 conjugate to CD10+ tumors was significantly blocked by NL-1 antibody.
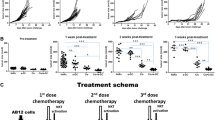
In vivo antitumor activity of the NL-1 conjugate was then tested against a CD10+ tumor transplanted to nude mice, and side effects were recorded. The NL-1 conjugate (4 mg/kg) showed anin vivo antitumor effect similar to MMC (2 mg/kg), which is at nearly maximal tolerable dose; the latter induced decreases in numbers of leukocytes and platelets, while the former did not, suggesting less side effect by the NL-1 conjugate. Since MMC demonstrates a broad spectrum of antitumor activity, the conjugate, as such, may be applicable for the treatment of cancer patients.
Similar content being viewed by others
Abbreviations
- ALL:
-
acute lymphocytic leukemia
- CML:
-
chronic myclocytic leukemia
- h:
-
hour
- IgG:
-
immunoglobulin G
- MMC:
-
mitomycin C
- MMC-D:
-
mitomycin C derivative
- PBS:
-
phosphate buffered saline
References
Usubuchi I, Sobajima Y, Hongo T, Kawaguchi T, Sugawara M, Matsui M, Wakaki S, Uzu K. Antitumor studies on mitomycin derivatives. 1. Effect on Hirosaki ascites sarcoma. Gann 1967; 58: 307–13.
Kinoshita S, Uzu K, Nakano K, Shimizu M, Takahashi T, Matsui M. Mitomycin derivatives. I. Preparation of mitosane and mitosene compounds and their biological activities. J Med Chem 1971; 14: 103–9.
Iyengar BS, Lin H-J, Cheng L, Remers WA. Development of new mitomycin C and porfiromycin analogues. J Med Chem 1981; 24: 975–81.
Iyengar BS, Sami SM, Remers WA, Bradner WT, Schurig JE. Mitomycin C and porfiromycin analogues with substituted ethylamines at position 7. J Med Chem 1983; 26: 16–20.
Iyengar BS, Sami SM, Tarnow SE, Remers WA, Bradner WT, Schurig JE. Mitomycin C analogues with secondary amines at position 7. J Med Chem 1983; 26: 1453–7.
Sami SM, Iyengar BS, Tarnow SE, Remers WA, Bradner WT, Schurig JE. Mitomycin C analogues with aryl substituents on the 7-amino group. J Med Chem 1984; 27: 701–8.
Imai R, Morimoto M. Comparative antitumor activities of 7-N-(p-hydroxyphenyl)mitomycin C (M-83) and mitomycin C. J Antibiot (Tokyo) 1983; 36: 559–65.
Morimoto M, Ashizawa T, Ohno H, Azuma M, Kobayashi E, Okabe M, Gomi K, Kono M, Saitoh Y, Kanda Y, Arai H, Sato A, Kasai M, Tsuruo T. Anti-tumor activity of 7-N-{{2-{[2-(γ-L-glutamylamino)ethyl] dithio}}ethyl}} mytomycin
Kono M, Saitoh Y, Kasai M, Sato A, Shirahata K, Morimoto M, Ashizawa T. Synthesis and antitumor activity of a novel water soluble mitomycin analog; 7-N{{2-{[2-(γ-L-glutamylamino)ethyl]dithio)}mitomycin C. Chem Pharm Bull (Tokyo) 1989; 37: 1128–30.
Taylor, PW, Howes C. Rationale for targeted drug delivery. Biotherapy 1991; 3: 1–8.
Foon KA, Bernhard MI, Oldlam RK Monoclonal antibody therapy: Assessment by animal tumor models. J Biol Res Mod 1982; 1: 277–304.
Ghose T, Blair AH. The design of cytotoxic-agent antibody conjugates. Crit Rev Ther Drug Carrier Syst 1987; 3: 263–359.
Ghose TI, Blair AH, Kulkarini PN. Preparation of antibody-linked cytotoxic agents. Methods Enzymol 1983; 93: 280–333.
Endo N, Takeda Y, Umemoto N, Kishida K, Watanabe K, Satio M, Kato Y, Hara T. Nature of Linkage and mode of action of methotrexate conjugated with antitumor antibodies: implications for future preparation of conjugates. Cancer Res 1988; 48: 3330–5.
Braslawsky GR, Eddson MA, Peace W, Kaneko T, Greenfield RS. Antitumor activity of adriamycin(hydrazone-linked) immunoconjugates compared with free adriamycin and specificity of tumor cell killing. Cancer Res 1990; 50: 6600–7.
Suzuki T, Sato E, Goto K, Katsurada Y, Unno K, Takahashi T. The preparation of mycomycin C, adriamycin and daunomycin covalently bound to antibodies as improved cancer chemotherapeutic agents. Chem Pharm Bull 1981; 29: 844–8.
Kato Y, Tsukada Y, Hara T, Hirai H. Enhanced antitumor activity of mitomycin C conjugated with anti-α fetoprotein antibody by a novel method of conjugation. J Appl Biochem 1983; 5: 313–9.
Umemoto N, Kato Y, Takeda Y, Saito M, Hara T, Seto M, Takahashi T. Conjugates of mitomycin C with the immuno-globulins M monomer fragment of a monoclonal anti-MM46 immunoglobulin M antibody with or without serum albumin as intermediary. J Appl Biochem 1984; 66: 297–307.
Manabe Y, Tsubota T, Haruta Y, Kataoka K, Okazaki M, Haisa S, Nakamura K, Kimura I. Production of a monoclonal antibody-mitomycin C conjugate, utilizing dextran T-40 and its biological activity. Biochem Pharmacol 1985; 34: 289–91.
Dillman RO, Johnson DE, Ogden J, Beidler D. Significance of antigen, drug, and tumor cell targets in the preclinical evaluation of doxorubicin, daunomycin, methotrexate, and mitomycin C monoclonal antibody immunoconjugates. Mol Biother 1989; 1: 250–5.
Shida Y, Ashizawa T, Morimoto M, Kuroda T. USP 4, 863,935 Sep, 5, 1989.
Ueda R, Tanimoto M, Takahashi T, Ogata S, Nishida K, Namikawa R, Nishizuka Y, Ota K. Serological analysis of cell acute lymphocytic leukemia by mouse monoclonal antibodies. Proc Natl Acad Sci USA 1982; 79: 4386–90.
Geran RI, Greenberg NH, MacDonald MM, Schumacher AM, Abbott BJ. Protocol for screening chemical and natural products against animal tumors and other biological systems. Cancer Chemother Rep part III 1972; 3: 1–103.
Ehrlich PA. A general review of the recent work in immunity. In: The collected works of Paul Ehrlich. New York: Pergamon Press, 1956: 442–7.
Seto M, Umemoto N, Saito M, Mazuko Y, Hara T, Takahashi T. Monoclonal anti-MM4 antibody: ricin A chain conjugate:in vitro andin vivo antitumor activity. Cancer Res 1982; 42: 5209–15.
Vitetta ES, Fulton RJ, May RD, Till M, Uhr, JW. Redesigning nature's poisons to create anti-tumor reagents. Science 1987; 238: 1098–104.
Filipovich AH, Vallora DA, Youle RJ, Quinones RR, Neville DM, Kersey JH.Ex-vivo treatment of donor bone marrow with anti-T cell immunotoxins for prevention of graft-versus-host disease. Lancet 1984; 1: 469–71.
Press OW. Immunotoxins. Biotherapy 1991; 3: 65–76.
Eichler H-G. Clinical experience of targeted therapy. Biotherapy 1991; 3: 77–85.
Takahashi T, Yamaguchi T, Kitamura K, Suzuyama H, Honda M, Yokota T, Kotanagi H, Takahashi M, Hashimoto Y. Clinical application of monoclonal antibody-drug conjugates for immunotargeting chemotherapy of colorectal carcinoma. Cancer 1988; 61: 881–8.
Tjandra JJ, Pietersz GA, Teh JG, Euthbertson AM, Sullivan JR, Penfold C, McKenzie IFC. Phase I clinical trial of drug-monoclonal antibody conjugates in patients with advanced colorectal carcinoma: A preliminary report. Surgery 1989; 116: 533–45.
Shida Y., manuscript in preparation.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shida, Y., Okabe, M., Kuroda, T. et al. In vivo andin vitro antitumor activity of mitomycin C conjugates at 7-N position through a linker containing thiocarbamate bond with CD10 monoclonal antibody. Biotherapy 5, 97–105 (1992). https://doi.org/10.1007/BF02171694
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02171694