Abstract
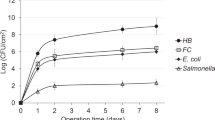
A near-shore coastal mangrove island receiving untreated sewage and a coastal cove receiving rum distillery effluent in Puerto Rico were examined for their ability to support survival and activity ofKlebsiella pneumoniae andEscherichia coli. Pure cultures of both bacteria were monitored for 96 hours in situ at both locations using membrane diffusion chambers.K. pneumoniae survived at all sites as measured by AODC and Coulter Counter direct counts. However, at the mangrove island less than 20% of theK. pneumoniae population was active (AODC) after the first 3 hours and less than 10% of this population was respiring (INT). In contrast, the coastal area which was receiving rum distillery effluent was able to maintain 40% of theK. pneumoniae population in an active state with 90% respiring. TheE. coli population declined by two orders of magnitude at the mangrove island, but remained unchanged at the rum distillery outfall. TheE. coli population had a higher proportion of active cells and respiring cells thanK. pneumoniae at all sites. At the rum distillery site, theE. coli population was remarkable in that 95% remained active and 99% were respiring. This study suggests that, when sufficient organic loading exists,E. coli, a “nonsurvivor,” can overcome the bactericidal effects of tropical marine waters.K. pneumoniae, a “survivor,” could survive under all conditions but could not maintain the activity or respiration that theE. coli population could, even when high organic loads were present. Morphological changes related to nutrient stress in the tropical marine environment were apparent inE. coli, but not inK. pneumoniae. Based on physiological activityE. coli is just as much a “survivor” asK. pneumoniae in tropical marine waters.
Similar content being viewed by others
References
Almy R, Carrion L (1963) Shallow water stony corals of Puerto Rico. Carib J Sci 3:133–162
American Public Health Association Standard Method for the Examination of Water and Wastewater (1981) 15th ed. American Public Health Association, Washington, DC
Bagley ST, Seidler RJ (1977) Significance of fecal coliform-positiveKlebsiella. Appl Environ Microbiol 33:1141–1148
Baker RM, Singleton FL, Hood MA (1983) Effects of nutrient deprivation onVibrio cholerae. Appl Environ Microbiol 46:930–940
Biamon EJ, Hazen TC (1983) The distribution and survival ofAeromonas hydrophila in tropical near-shore coastal waters receiving rum distillery effluent. Water Res 17:319–326
Bonde GJ (1977) Bacterial indication of water pollution. Adv Aquatic Microbiol 1:327–450
Brown C, Seidler RJ (1973) Potential pathogens in the environment:Klebsiella pneumoniae, a taxonomic and ecological enigma. Appl Microbiol 25:900–904
Cabelli VJ, Dufour AP, McCabe LJ, Levin MA (1983) A marine recreational water quality criterion consistent with indicator concepts and risk analysis. J Water Pollut Cont Fed 55:1306–1314
Campbell L, Roth RL (1975) Methyl violet: a selective agent for differentiation ofKlebsiella pneumoniae fromEnterobacter aerogenes and other gram-negative organisms. Appl Microbiol 24:258–261
Carrillo M, Estrada E, Hazen TC (1985) Survival and enumeration of the fecal indicatorsBifidobacterium adolescentis andEscherichia coli in a tropical rain forest watershed. Appl Environ Microbiol 50:468–476
Costle DM (1979) Effect of distillery wastes on the marine environment. US Environmental Protection Agency, Washington, DC
Daley RJ (1979) Direct epifluorescence enumeration of native aquatic bacteria: uses, limitations, and comparative accuracy. In: Costerton JW, Colwell RR (eds) Native aquatic bacteria: enumeration, activity, and ecology. American Society for Testing and Materials, Philadelphia
Duncan DW, Razzell WE (1972)Klebsiella biotypes among coliforms isolated from forest environments and farm produce. Appl Microbiol 24:933–938
Fuentes FA, Biamon EJ, Hazen TC (1983) Bacterial chemotaxis to effluent from a rum distillery in tropical near-shore coastal waters. Appl Environ Microbiol 46:1438–1441
Fuentes FA, Hazen TC, Lopez-Torres AJ, Rechani P (1985)Klebsiella pneumoniae in orange juice concentrate. Appl Environ Microbiol 49:1527–1530
Gonzalez JG, Yoshioka PM, Zimmerman RJ, Lopez JM, Hernandez-Avila M, Suhayda JN, Roberts HH, Cruz Baez D, Pesante D, Velazco AT (1979) Biological effects of rum slops in the marine environment. US Environmental Protection Agency, Washington, DC
Hazen TC, Aranda CF (1981) Bacteria and water quality in the Rio Mameyes watershed. In: Seventh Natural Resources of Puerto Rico Symposium. Department of Natural Resources, Commonwealth of Puerto Rico, San Juan, pp 87–111
Hazen TC, Esch GW (1983) Effect of effluent from a nitrogen fertilizer factory and a pulp mill on the distribution and abundance ofAeromonas hydrophila in Albemarle Sound North Carolina. Appl Environ Microbiol 45:31–42
Hazen TC, Prieto L, Lopez-Torres AJ, Biamon E (1982) Survival and activity of fecal coliform bacteria in near-shore coastal waters. In: Eighth Natural Resources of Puerto Rico Symposium. Department of Natural Resources. Commonwealth of Puerto Rico, San Juan, pp 128–161
Hobbie JE, Daley RJ, Jasper S (1977) Use of Nuclepore filters for counting bacteria by fluorescence microscopy. Appl Environ Microbiol 33:1225–1228
Jones KL, Rhodes-Roberts ME (1981) The survival of marine bacteria under starvation conditions. J Appl Bacteriol 50:247–258
Knittel MD (1975) Occurrence ofKlebsiella pneumoniae in surface waters. Appl Microbiol 29:595–597
Knittel MD, Seidler RJ, Eby C, Cabe LM (1977) Colonization of the botanical environment byKlebsiella isolates of pathogenic origin. Appl Environ Microbiol 34:557–563
Lavoie MC (1983) Identification of strain isolates as total and fecal coliforms and comparison of both groups as indicators of fecal pollution in tropical climates. Can J Microbiol 29:689–693
Matsen JM, Spindler JA, Blosser RO (1974) Characterization ofKlebsiella isolates from natural receiving waters and comparison with human isolates. Appl Microbiol 28:672–678
McFeters GA, Stuart DG (1972) Survival of coliform bacteria in natural waters: field and laboratory studies with membrane-filter chambers. Appl Microbiol 24:805–811
Niemela SI, Vaatanen P (1982) Survival in lake water ofKlebsiella pneumoniae discharged by a paper mill. Appl Environ Microbiol 44:264–269
Seidler RJ, Knittel MD, Brown C (1975) Potential pathogens in the environment cultural reactions and nucleic acid studies onKlebsiella pneumoniae from clinical and environmental sources. Appl Microbiol 29:819–825
Seidler RJ, Morrow E, Bagley ST (1977) Klebsielleae in drinking water emanating from redwood tanks. Appl Environ Microbiol 33:893–900
Sjogren RE, Gibson MJ (1981) Bacterial survival in a dilute environment. Appl Environ Microbiol 41:1331–1336
Taylor SL, Guthertz LS, Leatherwood M, Lieber ER (1976) Histamine production ofKlebsiella pneumoniae and an incident of scombroid fish poisoning. Appl Environ Microbiol 37:274–278
US Water Resources Council Caribbean Region (1978) US Water Resources Council, Washington, DC
Vasconcelos GJ, Swartz RG (1976) Survival of bacteria in seawater using a diffusion chamber apparatus in situ. Appl Environ Microbiol 31:913–920
Xu H-S, Roberts N, Singleton FL, Attwell RW, Grimes DJ, Colwell RR (1982) Survival and viability of nonculturableEscherichia coli andVibrio cholerae in the estuarine and marine environment. Microb Ecol 8:313–323
Zar JH (1984) Biostatistical analysis. Prentice-Hall Inc, Englewood Cliffs, NJ
Zimmerman R, Iturriaga R, Becker-Birck J (1978) Simultaneous determination of the total number of aquatic bacteria and the number thereof involved in respiration. Appl Environ Microbiol 36:926–934
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lopez-Torres, A.J., Prieto, L. & Hazen, T.C. Comparison of the in situ survival and activity ofKlebsiella pneumoniae andEscherichia coli in tropical marine environments. Microb Ecol 15, 41–57 (1988). https://doi.org/10.1007/BF02012951
Issue Date:
DOI: https://doi.org/10.1007/BF02012951