Abstract
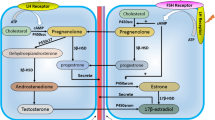
Thein vitro secretion of 17α,20β-dihydroxy-4-pregnen-3-one 20-sulphate (17α,20β-P-sulphate) and the free steroid 17α,20β-dihydroxy-4-pregnen-3-one (17α,20β-P), by rainbow trout (Oncorhynchus mykiss) gonads, in response to gonadotropin (GTH) I and GTH II, were studied during the final stages of sexual maturation. Substantial amounts of 17α,20β-P-sulphate were produced, by both mature ovaries and testes, indicating considerable 20β-hydroxysteroid sulphotransferase (20β-HST) activity within these tissues. In the post-ovulatory ovary the level of 17α,20β-P-sulphate (36.6 ng ml−1) greatly exceeded that of 17α,20β-P (8.59 ng ml−1). The amount of 17α,20β-P-sulphate produced in incubations of both mature ovary and testes was unaffected by either GTH I or GTH II treatment at physiological concentrations up to 100 ng ml−1. Similarly, incubations of maturing ovary and testes, treated with GTH I or GTH II, in the presence of added 17α,20β-P at 100 ng ml−1 of medium, produced levels of 17α,20β-P-sulphate that were similar to those of the controls. In incubations of mature ovarian follicles at the stages of germinal vesicle breakdown and preovulation, both GTHs significantly stimulated secretion of 17α,20β-P, although GTH II was always more potent than GTH I. GTH II significantly elevated the levels of 17α,20β-P in testicular incubations from mature males more than 4-fold relative to GTH I and controls, which did not differ from one another.
In conclusion, 20β-HST, the enzyme responsible for the sulphate conjugation of 17α,20β-P, was found to be active in the ovaries and testes of rainbow troutin vitro. However, the levels of this enzyme do not appear to be regulated by either GTH I or GTH II.
Similar content being viewed by others
References cited
Dickhoff, W.W. and Swanson, P. 1990. Functions of salmon pituitary glycoprotein hormones: “The maturational surge hypothesis”.In Progress in Comparative Endocrinology. pp. 349–356. Edited by A. Epple, C.G. Scanes and M.H. Stetson. Wiley-Liss, Inc., New York.
Kanamori, A., Adachi, S. and Nagahama, Y. 1988. Developmental changes in steroidogenic responses of ovarian follicles of amago salmon (Oncorhynchus rhodurus) to chum salmon gonadotropin during oogenesis. Gen. Comp. Endocrinol. 72: 13–24.
Miwa, S., Yan, L. and Swanson, P. 1994. Localization of two gonadotropin receptors in the salmon gonad byin vitro ligand autoradiography. Biol. Reprod. 50: 629–642.
Moore, A. and Scott, A.P. 1992. 17α,20β-dihydroxy-4-pregnen-3-one 20-sulphate is a potent odorant in precocious male Atlantic salmon (Salmo salar L.) parr which have been pre-exposed to urine of ovulated females. Proc. Roy. Soc. Lond. 249B: 205–209.
Nagahama, Y. and Adachi, S. 1985. Identification of a maturation-inducing steroid in a teleost, the amago salmon (Oncorhynchus rhodurus). Dev. Biol. 109: 428–435.
Planas, J.V., Swanson, P. and Dickhoff, W.W. 1993. Regulation of testicular steroid productionin vitro by gonadotropins (GTH I and GTH II) and cyclic AMP in coho salmon (Oncorhynchus kisutch). Gen. Comp. Endocrinol. 91: 8–24.
Planas, J.V. and Swanson, P. 1995. Maturation-associated changes in the response of salmon testis to the steroidogenic actions of gonadotropins (GTH I and GTH II)in vitro. Biol. Reprod. 52: 697–704.
Scott, A.P., Sheldrick, E.L. and Flint, A.P.F. 1982. Measurement of 17α,20β-dihydroxy-4-pregnen-3-one in plasma of trout (Salmo gairdneri Richardson): seasonal changes in response to salmon pituitary extract. Gen. Comp. Endocrinol. 46: 444–451.
Scott, A.P. and Canario, A.V.M. 1992. 17α,20β-dihydroxy-4-pregnen-3-one 20-suphate: a major metabolite of the oocyte maturation-inducing steroid. Gen. Comp. Endocrinol. 85: 91–100.
Scott, A.P. and Turner, R.J. 1991. 17α,20β-dihydroxy-4-pregnen-3-one 20-sulphate: a major metabolite of the oocyte maturation-inducing steroid in plaice (Pleuronectes platessa) and rainbow trout (Oncorhynchus mykiss).In Proc. 4th Int. Symp. on Reproductive Physiology of Fish. p. 92. Edited by A.P. Scott, J.P. Sumpter, D.E. Kime and M.S. Rolfe. FishSymp 91. Sheffield.
Scott, A.P. and Liley, N.R. 1994. Dynamics of excretion of 17α,20β-dihydroxy-4-pregnen-3-one 20-sulphate, and of the glucuronides of testosterone and 17β-oestradiol, by urine of reproductively mature male and female rainbow trout (Oncorhynchus mykiss). J. Fish Biol. 44: 117–129.
Scott, A.P., Liley, N.R. and Vermeirssen, E.V.M. 1994. Urine of reproductive mature female rainbow trout,Oncorhynchus mykiss (Walbaum), contains a priming pheromone which enhances levels of sex steroids and gonadotropin II in males. J. Fish Biol. 44: 131–147.
Scott, A.P., Nagahama, Y., Van Der Kraak, G. and Nagler, J.J. 1995. Sulphation and uptake of the maturation-inducing steroid, 17α,20β-dihydroxy-4-pregnen-3-one, by rainbow trout ovarian follicles. Fish Physiol. Biochem 14: 301–311.
Scott, A.P. and Sorenson, P.W. 1994. Time course of release of pheromonally active gonadal steroids and their conjugates by ovulatory goldfish. Gen. Comp. Endocrinol. 96: 309–323.
Scott, A.P. and Vermeirssen, E.V.M. 1994. Production of conjugated steroids by teleost gonads and their role as pheromones.In Perspectives in Comparative Endocrinology. pp. 645–654. Edited by K.G. Davey, R.E. Peter and S.S. Tobe. National Research Council of Canada, Ottawa, Canada.
So, Y.P., Idler, D.R., Truscott, B. and Walsh, J.M. 1985. Progestogens, androgens and their glucuronides in the terminal stages of oocyte maturation in landlocked Atlantic salmon. J. Steroid Biochem. 23: 583–591.
Sumpter, J.P. and Scott, A.P. 1989. Seasonal variations in plasma and pituitary levels of gonadotrophin in males and females of two strains of rainbow trout (Salmo gairdneri). Gen. Comp. Endocrinol. 75: 376–388.
Suzuki, K., Nagahama, Y. and Kawauchi, H. 1988a. Steroidogenic activities of two distinct salmon gonadotropins. Gen. Comp. Endocrinol. 71: 452–458.
Suzuki, K., Kanamori, A., Nagahama, Y. and Kawauchi, H. 1988b. Development of salmon GTH I and GTH II radioimmunoassays. Gen. Comp. Endocrinol. 71: 459–467.
Swanson, P. 1991. Salmon gonadotropins: reconciling old and new ideas.In Proc. 4th Int. Symp. on Reproductive Physiology of Fish. p. 2. Edited by A.P. Scott, J.P. Scott, J.P. Sumpter, D.E. Kime and M.S. Rolfe. FishSymp 91, Sheffield.
Yan, L., Swanson, P. and Dickhoff, W.W. 1992. A two-receptor model for salmon gonadotropins (GTH I and GTH II). Biol. Reprod. 47: 418–427.
Young, G., Kagawa, H. and Nagahama, Y. 1982. Oocyte maturation in the amago salmon(Oncorhynchus rhodurus): in vitro effects of salmon gonadotropin, steroids, and cyanoketone (an inhibitor of 3β-hydroxysteroid dehydrogenase). J. Exp. Zool. 224: 265–275.
Van Der Kraak, G. and Donaldson, E.M. 1986. Steroidogenic capacity of coho salmon ovarian follicles throughout the periovulatory period. Fish Physiol. Biochem. 1: 179–186.
Zar, J.H. 1984. Biostatistical Analysis. Prentice Hall Inc., Englewood Cliffs.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nagler, J.J., Scott, A.P., Tyler, C.R. et al. Gonadotropins I and II do not stimulate thein vitro secretion of 17α,20β-dihydroxy-4-pregnen-3-one 20-sulphate by rainbow trout gonads during final sexual maturation. Fish Physiol Biochem 15, 149–156 (1996). https://doi.org/10.1007/BF01875594
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01875594