Summary
The gravity coalescence of drops at flat fluid/liquid interfaces was studied in three-phase systems. Rest-time distributions were in qualitative agreement with the earlier theory of Charles and Mason for two-phase systems. The probability of coalescence increased inversely with the square of intervening film thickness. Partial coalescence was similar to that previously observed in two-phase systems. The viscosity of the drop affected the mechanism of coalescence more than the viscosities of the other phases.
Zusammenfassung
Es wurde die Koaleszenz von Tropfen an ebenen Flüssigkeits-Flüssigkeitsgrenzflächen in Dreiphasensystemen untersucht. Die beobachtete Verteilung der Verweilzeiten ist in qualitativer Übereinstimmung mit der Theorie von Charles und Mason für Zweiphasensysteme. Die Wahrscheinlichkeit dafür, daß Koaleszenz auftritt, nimmt umgekehrt proportional mit dem Quadrat der Filmdicke zwischen Tropfen und Phasengrenze zu. Die partielle Koaleszenz war ähnlich, wie sie früher in Zweiphasensystemen beobachtet worden war. Die Viskosität des Tropfens beeinflußt den Mechanismus der Koaleszenz mehr als die Viskosität der anderen Phasen.
Similar content being viewed by others
Abbreviations
- a i :
-
horizontal diameter of the i th generation drop
- B, H, N :
-
levels of the bottom, top and neck of the coalescing drop
- c :
-
radius of contact
- f(τ) :
-
fraction of rest-times ⩽ τ
- g :
-
acceleration due to gravity
- h 1/2 :
-
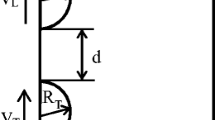
median thickness of the intervening film
- K III :
-
constant characterising the rate of thinning of the intervening film in three-phase systems (eq.[1b])
- p=η 1/η 2 :
-
viscosity ratio
- R :
-
radius of hole in the intervening film
- R 0 :
-
radius of neck in the outflowing column of phase 1
- S 1=σ 23−(σ 12+σ 13):
-
spreading coefficient of phase I
- t :
-
time
- Ū v :
-
average vertical velocity of flow through the neck
- V 1 :
-
volume of drop (phase 1)
- δ i , η i :
-
density, viscosity of phase i
- v i :
-
kinematic viscosity of phase i
- σ ij :
-
interfacial tension between phases i and j
- τ; τ 1/2, τ max, τ min :
-
rest-time; median, maximum and minimum rest-times
References
Cockbain, E. G. and T. S. McRoberts, J. Colloid Sci. 8, 440 (1953).
Gillespie, T. S. and E. K. Rideal, Trans. Faraday 52, 173 (1956).
Elton, G. A. H. and R. G. Picknett, 2nd International Congress on Surface Activity, Proceedings 1, 287 (London 1957).
Nielsen, E., R. Wall, and G. Adams, J. Colloid Sci. 13, 441 (1958).
Charles, G. E. and S. G. Mason, J. Colloid Sci. 15, 236 (1960).
Allan, R. S. and S. G. Mason, Trans. Faraday 57, 2027 (1961).
MacKay, G. D. M. and S. G. Mason, Can. J. Chem. Eng. 41, 203 (1963).
Charles, G. E. and S. G. Mason, J. Colloid Sci. 15, 105 (1960).
Princen, H. M. and S. G. Mason, J. Colloid Sci. 20, 246 (1965).
Harkins, W. D., Physical Chemistry of Surface Films (New York 1952).
Mar, A., M. Sc. Thesis, McGill University (Montreal 1967).
Mar, A. and S. G. Mason, Part II. (Following Paper).
Hodgson, T. D. and J. C. Lee, Preprint 28 C, International Symposium on Coalescence, 59th National Meeting, A.I.Ch.E. (New York 1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mar, A., Mason, S.G. Coalescence in three-phase fluid systems. Kolloid-Z.u.Z.Polymere 224, 161–172 (1968). https://doi.org/10.1007/BF01533973
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01533973