Abstract
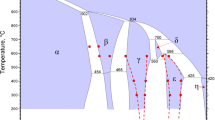
From co-precipitated powder samples, the solid state reactions occurring between room temperature and 1500° C in the ZrO2-CaO system have been studied. At low temperatures, compositions containing < 25 mol% CaO show a complex picture of phase transformation and ordering in the system. From the obtained results the following singular reactions have been established. (i) Tetragonal zirconia solid solution decomposes eutectoidally at 7 mol% CaO and 1048 ± 4° C into monoclinic zirconia solid solution and calcium zirconate (CZ). (ii) Cubic zirconia solid solution undergoes a eutectoidal decomposition at 17.5 mol% Cao and 1080 ±20° C into tetragonal solid solution + calcium zirconate. (iii) The monoclinic ordered phase, CaZr4O9 (Φ1), ), undergoes an order-disorder transformation into cubic zirconia solid solution at 1232 ± 5° C. (iv) Cubic zirconia solid solution undergoes a eutectoidal decomposition into two ordered phases, Φ1 + Φ2 at 21 mol% CaO and 1200 ± 10°C. (v) Hexagonal ordered phase Ca6Zr19O44 (Φ2) decomposes peritectoidally into cubic zirconia solid solution + calcium zirconate at 1360 ± 10° C. The two ordered phases Φ1 and Φ2 seem to be unstable below ≈ 1100° C. By using DTA, X-ray diffraction and SEM techniques, the extent of the tetragonal and cubic zirconia solid solution fields have been established. From the above experimental results a new tentative phase diagram is given for the ZrO2-rich region of the system, ZrO2-CaO.
Similar content being viewed by others
References
P. Duwez, F. Odell andF. H. Brown Jr,J. Amer. Ceram. Soc. 35 (1952) 107.
A. Dietzel andH. Tober,Ber. Deut. Keram. Ges. 30 (1953) 71.
B. C. Weber, H. J. Garret, F. A. Maurer andM. A. Schwartz,J. Amer. Ceram. Soc. 39 (1956) 197.
S. Fernandes andL. Beaudin, presented at the 21st Annual Pittsburgh Diffraction Conference, Mellon Institute, Pittsburgh, Pennsylvania, November 1963.
R. Roy, H. Miyabe andA. M. Diness, presented at the 66th Annual Meeting of the American Ceramic Society, Chicago, April 21, 1964 (Basic Science Division No. 11-B-64); for abstract seeAmer. Ceram. Soc. Bull. 43 (1964) 255.
I. Barbariol,Ann. Chim. (Rome) 55 (1965) 321.
R. Garvie,J. Amer. Ceram. Soc. 51 (1968) 553.
V. S. Stubican andS. P. Ray,ibid. 60 (1977) 534.
J. R. Hellmann andV. S. Stubican,Mater. Res. Bull. 17 (1982) 459.
Idem, J. Amer. Ceram. Soc. 66 (1983) 260.
G. B. Senft andV. S. Stubican,Mater. Res. Bull. 18 (1983) 1163.
B. Hudson andP. T. Moseley,J. Solid State Chem. 19 (1976) 383.
L. H. Schoenlein, L. W. Hobbs andA. H. Heuer,J. Appl. Crystallogr. 13 (1980) 375.
J. M. Marder, T. E. Mitchell andA. H. Heuer,Acta Met. 31 (1983) 387.
T. K. Gupta, F. F. Lange andJ. H. Bechtold,J. Mater. Sci. 13 (1978) 1464.
R. H. J. Hannink, K. A. Johnston, R. T. Pascal andR. C. Garvie, in “Advances in Ceramics”, Vol. 3, “Science and Technology of Zirconia”, edited by A. H. Heuer and L. W. Hobbs (American Ceramic Society, 1980) p. 116.
B. G. Hyde andL. Eyring, in “Rare Earth Research”, Vol. 111, edited by L. Eyring (Gordon and Breach, New York, 1965) p. 623.
J. G. Allpress, H. J. Rossell andH. G. Scott,Mater. Res. Bull. 9 (1974) 455.
P. Duran andJ. M. Rodriguez, presented at 12th Journees d'Etude des Equilibres entre Phases, Bordeaux, 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Duran, P., Recio, P. & Rodriguez, J.M. Low temperature phase equilibria and ordering in the ZrO2-rich region of the system ZrO2-CaO. J Mater Sci 22, 4348–4356 (1987). https://doi.org/10.1007/BF01132028
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01132028