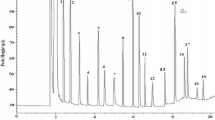
Abstract
Minor millets, viz. Barnyard millet, Proso millet, Little millet, Foxtail millet and Kodo millet, one variety in each grown in Tamil Nadu Agricultural University (TNAU), Coimbatore, Tamil Nadu were selected for the study. The protein contents of the selected decorticated millets were found to be 11.0, 12.3, 12.9, 10.5 and 10.6% respectively. Fractionation of these proteins revealed that prolamin forms major storage protein in Foxtail millet whereas, glutelin forms major storage protein in all the other millets. The extractability was studied using different solvents, viz. isopropyl alcohol, t-butyl alcohol and ethyl alcohol with varying concentration of 2-mercapto ethanol. Electrophoretic pattern of the extracted prolamins from these millets were compared and found that a protein band at the molecular weight range of 20 kD was found homologous in all except Proso millet. The extractability of the 20kD protein in 90% isopropyl alcohol showed its strong hydrophobic nature.
Similar content being viewed by others
References
Sudharshana L, Monterio PV, Ramachandra G (1988) Studies on the proteins of Kodo millet (Paspalum Scrobiculatum). J Sci Food Agric 42: 315–323.
Monterio PV, Virupaksha TK, Rao DR (1982) Proteins of Italian millet: Amono acid composition, solubility fractionation and electrophoresis of protein fractions. J Sci Food Agric 33: 1072–1079.
Monterio PV, Sudharshana L, Ramachandra G (1988) Japanese Barnyard millet (Echinochloa frumentacea) protein content, quality and SDS-PAGE of protein fractions. J Sci Food Agric 43: 17–25.
Burgess SR, Shewry PR (1986) Identification of homologous globulins from embryos of wheat, barley, rye and oats. J Exp Botany 37: 1863–1871.
Landry J, Moureaux T (1970) Hetero genetic des glutelins du grain de mais, extraction selective et composition en acids amines des trois fractions. Isolees Bull Soc Chem Biol 52: 1021–1037.
AOAC (1975) Official methods of analysis. Wasington, DC: Association of Official Analytical Chemists.
Harlow ED, Lane D (1988) Antibodies: A Laboratory manual. New York: Cold Spring Harbor Laboratory Publications, 670 pp.
Esen A, (1986) Separation of alcohol soluble proteins (Zeins) from maize into three fractions of differential solubility. Plant Physiol 80: 623–627.
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the bacteriophage T4. Nature 227: 680–685.
Ottoboni LMM, Leite A, Targon MLN, Crozier A, Arruda P (1990) characterization of the storage protein in seed ofCoix lacryma jobi var Adlay. J Agric Food Chem 38: 631–635.
Mazhar H, Chandrasekar A, Shetty HS (1993) Isolation and immunochemical characterization of the alcohol-extractable proteins (Kafirins) ofSorghum bicolor (L.) Moench. J Cereal Sci 17: 83–93.
Esen A, Conroy JM, Wang SZ (1983) A simple and dot immuno binding assay for Zein and other prolamins. Anal Biochemistry 132: 462–467.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Parameswaran, K.P., Thayumanavan, B. Homologies between prolamins of different minor millets. Plant Food Hum Nutr 48, 119–126 (1995). https://doi.org/10.1007/BF01088307
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01088307