Abstract
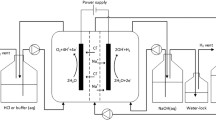
A hypochlorite cell divided by an anionic membrane was examined. The results clearly show that it is possible to suppress cathodic reduction of hypochlorite and to achieve a higher degree of chloride to hypochlorite conversion, at relatively high current efficiencies, than in the case of an undivided cell, using seawater or untreated diluted brine electrolyte.
Similar content being viewed by others
References
‘Local Generation and Use of Chlorine and Hypochlorite’, SCI Electrochemical Technology, Group Meeting, London, 14–16 October 1980.
R. Doan and A. Haimes, ‘Hypochlorite Generation at a Water Pollution Control Plant’,Public Works Magazine, January 1978.
A. F. Adamson, B. G. Lever and W. F. Stones,J. Appl. Chem. 13 (1963) 483.
J. E. Bennett,Chem. Eng. Prog. 70 (1974) 60.
A. T. Kuhn and R. B. Lartey,Chem. Ing. Techn. 47 (1975) 129.
G. H. Kelsall,J. Appl. Electrochem. 14 (1984) 177.
P. M. Robertson, M. Gnehm and L. Ponto,13 (1983) 307.
N. Ibl and D. Landolt,J. Electrochem. Soc. 115 (1968) 713.
N. Krstajić, V. Nakić and M. Spasojević,J. Appl. Electrochem. 17 (1987) 77.
L. L. Janssen and J. G. Hoogland,Electrochim. Acta 15 (1970) 1013.
M. Spasojević, N. Krstajić and M. Jakšić,Surf. Technol. 21 (1984) 19.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Krstajić, N., Nakić, V. & Spasojević, M. Hypochlorite production II. Direct electrolysis in a cell divided by an anionic membrane. J Appl Electrochem 21, 637–641 (1991). https://doi.org/10.1007/BF01024853
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01024853