Abstract
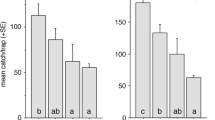
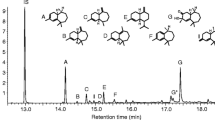
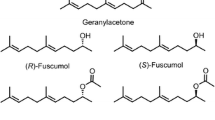
For two decades, the aggregation pheromone of the boll weevil,Anthonomus grandis Boh. (Coleoptera: Curculionidae), was thought to consist of four compounds: I [(+)-(Z)-2-isopropenyl-1-methylcyclobutane ethanol]; II [(Z)-3,3-dimethyl-ΔI,β-cyclohexane ethanol]; III [(Z)-3,3-dimethyl-Δ1,α-cyclohexane acetaldehyde); and IV [(E)-3,3-dimethyl-Δ1,α-cyclohexane acetaldehyde). Evidence is presented from behavioral and electrophysiological studies to show that only three of these components, I, II, and IV, are essential for attraction. Competitive field tests, in which each possible three-component blend was tested against the four-component mixture, demonstrated that omission of I, II. or IV resulted in decreased trap captures (P < 0.01). Trap captures by these blends lacking I, II, or IV resembled those by the hexane solvent alone in a similar experiment. However, omission of III did not significantly alter field attractiveness of the blend. Dosage-response curves constructed from electroantennogram responses of both males and females to serial dilutions of III, IV, and a 50∶50 mixture of the geometric isomers III and IV showed both sexes to be 10- to 100-fold more sensitive to IV than III. Data from the electrophysiological studies were consistent with a single acceptor type for the (E)-cyclohexylidene aldehyde, IV, for males, and possibly one or two acceptor types for III and IV for females. Possible roles for the (Z)-cyclohexylidene aldehyde, III, and implications for the pheromonal attractant currently used in boll weevil eradication/suppression programs are discussed.
Similar content being viewed by others
References
Brand, J.M. 1976. Bark beetle pheromones: Production of verbenone by a mycangial fungus ofDendroctonus frontalis.J. Chem. Ecol. 2:195–199.
Brand, J.M., Bracke, J.W., Markovetz, A.S.J., Wood, D.L., andBrowne, L.E. 1975. Production of verbenol pheromone by bacterium isolated from bark beetles.Nature 254:136–137.
Cross, W.H. andMitchell, H.C. 1966. Mating behavior of the boll weevil.J. Econ. Entomol. 59:1503–1507.
Cross, W.H. 1983. Ecology of cotton insects with special reference to the boll weevil, pp. 53–70,in R.L. Ridgway, E.P. Lloyd, and W.H. Cross (eds.). Cotton Insect Management with Special Reference to the Boll Weevil. USDA Handbook No. 589.
Cross, W.H., Lukefahr, M.J., Fryxell, P.A., andBurke, H.R. 1975. Host plants of the boll weevil.J. Econ. Entomol. 4:19–26.
Dickens, J.C. 1979. Electrophysiological investigations of olfaction in bark beetles.Mitt. Schweiz. Entomol. Ges. 52:203–216.
Dickens, J.C. 1981. Behavioural and electrophysiological responses of the bark beetle,Ips typographus, to potential pheromone components.Physiol. Entomol. 6:251–261.
Dickens, J.C. 1984. Olfaction in the boll weevil,Anthonomus grandis Boh. (Coleoptera: Curculionidae): Electroantennogram studies.J. Chem. Ecol. 10:1759–1785.
Dickens, J.C. 1986. Orientation of the boll weevil,Anthonomus grandis Boh. (Coleoptera: Curculionidae), to pheromone and volatile host compound in the laboratory.J. Chem. Ecol. 12:91–98.
Dickens, J.C., andBoldt, P.E. 1985. Electroantennogram responses ofTrirhabda bacharides (Weber) (Coleoptera: Chrysomelidae) to plant volatiles.J. Chem. Ecol. 11:767–779.
Dickens, J.C., andMoorman, E.E. 1987. Role of host plant and sensory environment in maturation of olfactory receptor system of the boll weevil,Anthonomus grandis.Soc. Neurosci. Abstr. 13:82.
Dickens, J.C., andMori, K. 1989. Receptor chirality and behavioral specificity of the boll weevil,Anthonomus grandis Boh. (Coleoptera: Curlionidae), for its pheromone, (+)-grandisol.J. Chem. Ecol. 15:517–528.
Dickens, J.C., Eischen, F.A., andDietz, A. 1986. Olfactory perception of the sex attractant pheromone of the greater wax moth,Galleria mellonella L. (Lepidoptera: Pyralidae), by the honey bee,Apis mellifera L. (Hymenoptera: Apidae).J. Entomol. Sci. 21:349–354.
Duncan, D.B. 1955. Multiple range and multiple F tests.Biometrics 11:1–42.
Glancey, B.M., andDickens, J.C. 1988. Behavioral and electrophysiological studies with larval rinses of the red imported fire ant,Solenopsis invicta Buren (Hymenoptera: Formidae).J. Chem. Ecol. 14:463–473.
Hardee, D.D., Cross, W.H., andMitchell, E.B. 1969. Male boll weevils are more attractive than cotton plants to boll weevils.J. Econ. Entomol. 62:165–169.
Hardee, D.D., Mckibben, G.H., Gueldner, R.C., Mitchell, E.B., Tumlinson, J.H., andCross, W.H. 1972. Boll weevils in nature respond to grandlure, a synthetic pheromone.J. Econ. Entomol. 65:97–100.
Hardee, D.D., Mckibben, G.H., Rummel, D.R., Huddleston, P.M., andCoppedoe, J.R. 1974. Response of boll weevils to component ratios and doses of the pheromone, grandlure.Environ. Entomol. 3:135–138.
Huddleston, P.M., Mitchell, E.B., andWilson, N.M. 1977. Disruption of boll weevil communication.J. Econ. Entomol. 70:83–85.
Kaissling, K.-E. 1971. Insect olfaction, pp. 351–431,in L. Beidler, (ed.). Handbook of Sensory Physiology, Vol. IV, Chemical Senses 1. Olfaction. Springer Verlag, New York.
Keller, J.C., Mitchell, E.B., Mckibben, G.H., andDavich, T.B. 1964. A sex attractant for female boll weevils from males.J. Econ. Entomol. 57:609–610.
Leufven, A., Bergstrom, G., andFalsen, E. 1986. Interconversion of verbenols and verbenone by identified yeasts isolated from the spruce bark beetleIps typographus.J. Chem. Ecol. 10:1349–1361.
Light, D.M., Jang, E.B., andDickens, J.C. 1988. Electroantennogram responses of the Mediterranean fruit fly,Ceratitis capitata, to a spectrum of plant volatiles.J. Chem. Ecol. 14:159–180.
Mckibben, G.H., Hardee, D.D., Davich, T.B., Gueldner, R.C., andHedin, P.A. 1971. Slowrelease formulations of grandlure, the synthetic pheromone of the boll weevil.J. Econ. Entomol. 64:317–319.
McKibben, G.H., Johnson, W.L., Edwards, R., Kotter, E., Kearny, J.F., Davich, T.B., Lloyd, E.P., andGanyard, M.C. 1980. A polyester-wrapped cigarette filter for dispensing grandlure.J. Econ. Entomol. 73:250–251.
Mitchell, E.B.,Hardee, D.D., andDavich, T.B. 1976. In-field boll weevil trap. U.S. Patent No. 3,949,515.
Mori, K., Tamada, S., andHedin, P.A. 1978. (−)-Grandisol, the antipode of the boll weevil pheromone, is biologically active.Naturwissenschaften 65:653–654.
Morse, D., andMeighen, E. 1986. Pheromone biosynthesis and role of functional groups in pheromone specificity.J. Chem. Ecol. 12:335–351.
Prestwich, G.D. 1987. Chemistry of insect hormone and pheromone metabolism.Science 237:999–1006.
Oakley, B., andSchafer, R. 1978. Experimental Neurobiology. The University of Michigan Press, Ann Arbor, xii + 367 pp.
Ostle, B. 1963. Statistics in Research. Iowa State University Press, Ames, Iowa. xv + 585 pp.
Ridgway, R.L., andLloyd, E.P. 1983. Evolution of cotton insect management in the United States, pp. 3–29,in R.L. Ridgway, E.P. Lloyd, and W.H. Cross (eds.). Cotton Insect Management with Special Reference to the Boll Weevil. USDA Handbook No. 589.
Tumlinson, J.H., Hardee, D.D., Gueldner, R.C., Thompson, A.C., Hedin, P.A., andMinyard, J.P. 1969. Sex pheromones produced by male boll weevil: Isolation, identification, and synthesis.Science 166:1010–1012.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dickens, J.C., Prestwich, G.D. Differential recognition of geometric isomers by the boll weevil,Anthonomus grandis Boh. (Coleoptera: Curculionidae): Evidence for only three essential components in aggregation pheromone. J Chem Ecol 15, 529–540 (1989). https://doi.org/10.1007/BF01014698
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01014698