Abstract
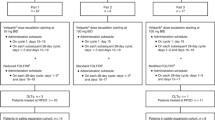
The ribonucleotide reductase inhibitor, hydroxyurea (HU), augments the cytotoxic effects of 5-fluorouracil (5FU)in vitro; both drugs are synergistic with interferon-α (IFN)in vitro. The aim of this phase I study was to determine the maximal duration of HU, 4.3 g/m2, administered as a parenteral infusion in combination with 5FU, 2.6 g/m2 administered over 24 hrs each week, + IFN, 9 MU, subcutaneously three times per week. There were 26 patients enrolled and evaluable. This included 14 patients with colorectal cancer of whom 13 had been previously treated, and 12 patients with other refractory malignancies (pancreas, cholangiocarcinoma, hepatocellular carcinoma, renal cell carcinoma, and others), of whom 10 were previously untreated. The dose-limiting toxicity of this regimen was myelosuppression. This prohibited dose escalation of HU above the starting dose (24 hrs) on a 6-weeks-on, 2-weeks-off therapy schedule. When filgrastim, 480 μg, was administered subcutaneously on days 3–6, the duration of HU could be extended to 48 hrs on a 2-weeks-on, 1 -week-off therapy schedule. There were two instances of fatal infection, one in a patient with a rectovaginal fistula with neutropenic sepsis and the second in a patient with non-neutropenicClostridium septicum sepsis. All therapy was administered in the ambulatory setting. There were three responders, all among previously untreated patients. High-dose parenteral hydroxyurea, 4.3 g/m2 administered over 24 hrs, can be safely combined with high-dose weekly 5FU, 2.6 g/m2 over 24 hrs + IFN, 9 MU subcutaneously three times per week, without filgrastim in the ambulatory setting. Parenteral hydroxyurea, 4.3 g/m2 over 24 hrs daily × 2 can also be combined with high-dose 5FU + IFN, but requires the addition of filgrastim to avoid severe myelosuppression.
Similar content being viewed by others
References
Stubbe J: Ribonucleotide reductases: amazing and confusing. J Biol Chem 265:5329–5332, 1990
Engstrom Y, Francke U: Assignment of the structural gene for subunit M1 of human ribonucleotide reductase to the short arm of chromosome 11. Exp Cell Res 158:477–483, 1985
Eriksson S, Graslund A, Skog S, Thelander L, Tribukait B: Cell-cycle dependent regulation of mammalian ribonucleotide reductase. J Biol Chem 259:11695–11700, 1984
Wright JA, McClarty GA, Lewis WH, Srinivasan PR: Hydroxyurea and related compounds. In: Gupta RS (ed), Drug Resistance in Mammalian Cells. CRC Press, Boca Raton, Fla, pp 15–27, 1989
Spears CP, Hayes A, Shahinian AHet al: Deoxyuridylate effects on thymidylate synthase-5-fluorodeoxyuridylate-folate ternary complex formation. Biochem Pharmacol 38:2985–2993, 1989
Moran RG, Danenberg PV, Heidelberger C: Therapeutic response of leukemic mice treated with fluorinated pyrimidines and inhibitors of deoxyuridylate synthesis. Biochem Pharmacol 31:2929–2935, 1982
Moran RG, Danenberg PV, Heidelberger C: Therapeutic response of leukemic mice treated with fluorinated pyrimidines and inhibitors of deoxyuridylate synthesis. Biochem Pharmacol 31:2929–2935, 1982
Muggia F, Moran RG: Treatment of colon cancer based on biochemical modulation of fluoropyrimidines by hydroxyurea. Semin Oncol 19 (suppl 9):90–93, 1992
Kobayashi S, Hoshino T: Combined cytotoxic effect of low-dose 5-fluorouracil and hydroxyurea on 9L cellsin vitro. Cancer Res 43:5309–5313, 1983
Hoshino T, Nagashima T: Factors modifying cytotoxicity induced by 5-FU and hydroxyurea. Cancer Treat Rep 69:993–997, 1985
Frankfurt OS: Enhancement of antitumor activity of 5-fluorouracil by drug combinations. Cancer Res 33:1043–1047, 1973
Lokich JJ, Pitman SW, Skarin AT: Combined 5-fluorouracil and hydroxyurea therapy for gastrointestinal cancer. Oncology 32:34–37, 1975
Lerner HJ: Clinical experience using hydroxyurea and 5-fluorouracil in hepatic metastasis from adenocarcinoma of the colon. Proc Am Assoc Cancer Res and Am Soc Clin Oncol 15:32, 1974
Walsh C, Speyer JL, Wernz JCet al.: Trial based on biochemical modulation of 5-fluorouracil (5FU) by daily sequential hydroxyurea (HU) in colorectal cancer. Proc Am Assoc Cancer Res 26:168, 1985
Kao AK, Muggia FM, Dubin Net al.: Evaluation of a sequential 5-FU and hydroxyurea combination in advanced bowel cancer. Cancer Treat Rep 68:1383–1385, 1984
Belt RJ, Haas CD, Kennedy J, Taylor S: Studies of hydroxyurea administered by continuous infusion: toxicity, pharmacokinetics, and cell synchronization. Cancer 46:455–162, 1980
Wadler S, Wersto R, Weinberg V, Thompson D, Schwartz EL: Interaction of fluorouracil and Interferon in human colon cancer cell lines: cytotoxic and cytokinetic effects. Cancer Res 50:5735–5739, 1990
National Cancer Institute: Guidelines for Reporting of Adverse Drug Reactions. Bethesda, MD, Division of Cancer Treatment, National Cancer Institute, 1988
Simon RM: Design and conduct of clinical trials. In: DeVita VT, Hellman S, Rosenberg S (eds). Cancer: Principles and Practice of Oncology, 3rd ed. New York, JB Lippincott Co, 1989, pp 396–420
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP: Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5:649–655, 1982
Kolbeinsson ME, Holder WD, Aziz S: Recognition, management, and prevention ofClostridium septicum abscess in immunosuppressed patients. Arch Surg 126:642–645, 1991
Alpern RJ, Dowell VR:Clostridium septicum infections and malignancy. J Am Med Assoc 209:385–388, 1969
Koransky JR, Stargel MD, Dowell VR:Clostridium septicum bacteremia: its clinical significance. Am J Med 66:63–66, 1979
Meropol NJ, Miller LJ, Korn EL, Braitman LE, MacDermott ML, Schucter LM: Severe myelosuppression resulting from concurrent administration of granulocyte colony-stimulating factor and cytotoxic chemotherapy. J Natl Cancer Inst 84:1201–1203, 1992
Yen Y, Zhou BS, Doroshow J: Low dose hydroxyurea synergy with 5-fluorouracil by modulating ribonucleotide reductase and thymidylate synthetase expression. Proc Am Assoc Cancer Res 35:329, 1994
Zhou BS, Cheng YC, Yen Y: Hydroxyurea resistant human KB cells exhibit collateral sensitivity to 5-fluorouracil under low dose hydroxyurea induction. Proc Am Assoc Cancer Res 35:329, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wadler, S., Haynes, H., Schechner, R. et al. Phase I trial of high-dose infusional hydroxyurea, high-dose infusional 5-fluorouracil and recombinant interferon-α-2a in patients with advanced malignancies. Invest New Drugs 13, 315–320 (1995). https://doi.org/10.1007/BF00873137
Issue Date:
DOI: https://doi.org/10.1007/BF00873137