Abstract
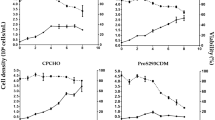
The effect of serum type and concentration on recombinant protein expression in vaccinia virus infected HeLa S3 cells was studied in both static and suspension culture. A model heterologous protein,β-galactosidase (β-gal), was used. Calf and horse sera in the range of 0.5–10%(v/v) were investigated. In static culture, the calf serum concentration did not show any significant influence on the β-gal production which was almost completed within 24h postinfection (pi). Higher horse serum concentration, on the other hand, resulted in higher β-gal concentration which continued to increase until 48 h pi. Total β-gal concentrations in 0.5% calf serum at 24 h pi and 10% horse serum at 48 h pi were 2.2±0.7 and 2.2±0.1 IU/ml, respectively. In suspension culture, both sera showed their respective effects on the β-gal production similar to those observed in static culture, indicating that the cultivation method had little influence on β-gal production. Accordingly, the use of 0.5% calf serum after virus infection in recommended for economical β-gal production.
Similar content being viewed by others
References
Barrett N, Mitterer A, Mundt W, Eibl J, Eibl M, Gallo RC, Moss B & Dorner F (1989) Large-scale production and purification of a vaccinia recombinant-derived HIV-1 gp160 and analysis of its immunogenicity. AIDS Res. Hum. Retroviruses 5: 159–171.
Blasco R & Moss B (1992) Role of cell-associated enveloped vacinia virus in cell-to-cell spread. J. Virol. 66: 4170–4179.
Buller RML & Palumbo GJ (1991) Poxvirus pathogenesis. Microbiol. Rev. 55: 80–122.
Chakrabarti S, Brechling K & Moss B (1985) Vaccinia virus expression vector: coexpression ofβ-galactosidase provides visual screening of recombinant virus plaques. Mol. Cell. Biol. 5: 3403–3409.
Chang A & Metz DH (1976) Further investigations on the mode of entry of vaccinia virus into cells. J. Gen. Virol. 32: 275–282.
Chillakuru RA, Ryu DDY & Yilma T (1991) Propagation of recombinant vaccinia virus in HeLa cells: adsorption kinetics and replication in batch cultures. Biotechnol. Prog. 7: 85–92.
Earl PL, Cooper N & Moss B (1991) Preparation of cell cultures and vaccinia virus stocks. In: Jassen K (ed.) Current protocols in molecular biology. Vol. 2 (pp. 16.16.1–16.16.7) John Wiley & Sons, New York.
Falkner FG, Turecek PL, MacGillivray RT, Bodemer W, Scheiflinger F, Kandels S, Mitterer A, Kistner O, Barrett N, Eibl J & Dorner F (1992) High level expression of active human prothrombin in a vaccinia virus expression system. Throm. Haemost. 68: 119–124.
Janeczko RA, Rodriguez JF & Esteban M (1987) Studies on the mechanism of entry of vaccinia virus in animal cells. Arch. Virol. 92: 135–150.
Mackett M (1991) Manipulation of vaccinia virus vectors. In: Murray EJ (ed.) Methods in molecular biology. Vol. 7, Gene transfer and expression protocols. (pp. 129–146) The Humana Press, Clifton, New Jersey.
Miner JN & Hruby DE (1990) Vaccinia virus: a versatile tool for molecular biologists. Trends Biotechnol. 8: 20–25.
Moss B (1990a) Poxviridae and their replication. In: Fields BN, Knipe DM, Chanock RM, Hirsch MS, Melnick JL, Monath TP & Roizman B (eds.) Virology. Vol. 2 (pp. 2079–2111) Raven Press, New York.
Moss B (1990b) Regulation of vaccinia virus transcription. Annu. Rev. Biochem. 59: 661–688.
Moss B (1991) Vaccinia virus: a tool for research and vaccine development. Science 252: 1662–1667.
Mukhopadhyay A, Mukhopadhyay SN & Talwar GP (1994) Physiological factors of growth and susceptibility to virus regulating Vero cells for optimum yield of vaccinia and cloned gene product (β-hCG). J. Biotech. 36: 177–182.
Payne LG & Norrby E (1978) Adsorption and penetration of enveloped and naked vaccinia virus particles. J. Virol. 27: 19–27.
Pieniazek D, Pieniazek NJ, Macejak D & Luftig RB (1990) Enteric adenovirus 41 (Tak) requires low serum for growth in human primary cells. Virology 178: 72–80.
Rice CM, Franke CA, Strauss JH & Hruby DE (1985) Expression of Sindbis virus structural proteins via recombinant vaccinia virus: synthesis, processing, and incorporation into mature Sindbis virions. J. Virol. 56: 227–239.
Shin JH, Lee GM & Kim JH (1994) Comparison of cell disruption methods for determiningβ-galactosidase activity expressed in animal cells. Biotechnol. Tech. 8: 425–430.
Smith GL, Mackett M & Moss B (1983) Infectious vaccinia virus recombinants that express hepatitis B virus surface antigen. Nature 302: 490–495.
Tartaglia J & Paoletti E (1988) Recombinant vaccinia virus vaccines. Trends Biotechnol. 6: 43–46.
Thomas G, Herbert E & Hruby DE (1986) Expression and cell type-specific processing of human preproenkephalin with a vaccinia recombinant. Science 232: 1641–1643.
Wickham TJ, Davis T, Granados RR, Shuler ML & Wood HA (1992) Screening of insect cell lines for the production of recombinant proteins and infectious virus in the baculovirus expression system. Biotechnol. Prog. 8: 391–396.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jun, S.C., Lee, G.M., Chang, S.H. et al. Effect of serum type and concentration on the expression ofβ-galactosidase in recombinant vaccinia virus infected HeLa S3 cells. Cytotechnology 19, 153–159 (1995). https://doi.org/10.1007/BF00749770
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00749770