Summary
Though sialoprotein nature of alkaline phosphatase of certain mammalian organs has been suggested by biochemical investigations, no histochemical techniques have yet been applied to elucidate this concept. With this view, the alkaline phosphatase of stomach and intestine of a mollusc—Semperula maculata—was analysed histochemically to elucidate its sialoglycoprotein nature. The localisation of alkaline phosphatase and sialic acid was investigated by employing well known and standard histochemical techniques.
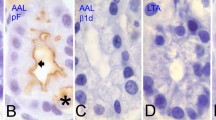
Alkaline phosphatase was localised selectively in the brush border of the mucosa of stomach and intestine, it was Mg++ nonsensitive but showed a structure-linked sensitivity to phenylalanine. The sialomucins were selectively localised in the brush border, whereas the goblet cells contained both the sialomucins and sulfomucins, and the connective tissue of lamina propria contained sulfomucins. The localisation of alkaline phosphatase and sialomucins in the brush border uniquely coincided with each other. The alkaline phosphatase activity in the brush border was completely lost after neuraminidase treatment at 37.5° C for 16 h. Such effect of neuraminidase on alkaline phosphatase activity was pH dependent and controlled by velocity of reaction. Heat-inactivated neuraminidase showed no effect on alkaline phosphatase activity.
These histochemical results have been interpreted as suggesting a sialoglycoprotein nature of alkaline phosphatase in the brush border, and sialic acid somehow seems to be essential for enzyme activity. These results, thus, indicate necessity of visualising some of the sialo-glycoproteins as macromolecules with catalytic activity.
Similar content being viewed by others
References
Butterworth, P. J., Moss, D. W.: Action of neuraminidase on human kidney alkaline phosphatase. Nature (Lond.) 209, 805–806 (1966).
Burstone, M. S.: Histochemical comparison of naphthol AS-phosphates for the demonstration of phosphatases. J. nat. Cancer Inst. 20, 601–615 (1958).
Curran, R. C.: The histochemistry of mucopolysaccharides. Int. Rev. Cytol. 17, 149–212 (1964).
Dempsey, E. W., Bunting, H., Singer, M., Wislocki, G. B.: The dye-binding capacity and other chemo-histological properties of mammalian mucopolysaccharides. Anat. Rec. 98, 417–429 (1947).
Fisher, E. R., Lillie, R. D.: The effect of methylation on basophilia. J. Histochem. Cytochem. 2, 81–87 (1954).
Fishman, W. H., Ghosh, N. K.: In: Advances in clinical chemistry, ed. by O. Bodansky and C. P. Stewart, vol X. New York: Academic Press 1967.
— Green, S., Inglis, N. I.: Organ specific behaviour exhibited by rat intestine and liver alkaline phosphatase. Biochim. biophys. Acta (Amst.) 62, 363–375 (1962).
—: L-phenylalanine: an organ specific, stereospecific inhibitor of human intestinal alkaline phosphatase. Nature (Lond.) 198, 685–686 (1963).
Ghosh, N. K., Goldman, S. S., Fishman, W. H.: Human placental alkaline phosphatase; a sialoglycoprotein. Enzymologia 33, 113–124 (1967).
Gomori, G.: Microscopic histochemistry. Principles and practice. Chicago: Chicago Univ. Press 1952.
Goreau, T.: Histochemistry of mucopolysaccharidelike substances and alkaline phosphatase in madreporaria. Nature (Lond.) 177, 1029–1030 (1956).
Grogg, E., Pearse, A. G. E.: A critical study of the histochemical techniques for acid phosphatase. J. Path. Bact. 64, 627–638 (1952).
Heilbronn, E.: Purification of cholinesterase from horse serum. Biochim. biophys. Acta (Amst.) 58, 222–230 (1962).
Holt, S. J.: Factors governing the validity of staining methods for enzymes and their bearing upon the Gomori acid phosphatase technique. Exp. Cell Res., Suppl. 7, 1–27 (1959).
Leppi, T. J.: Morphological analysis of mucous cells in the skin and slime glands of hagfish. Histochemie 15, 68–78 (1968).
Lev, R., Spicer, S. S.: Specific staining of sulphate groups with alcian blue at low pH. J. Histochem. Cytochem. 12, 309 (1964).
McManus, J. F. A.: Histological demonstration of mucin with periodic acid. Nature (Lond.) 158, 202 (1946).
Moog, F., Wegner, E. L.: The occurrence of a neutral mucopolysaccharide at sites of high alkaline phosphatase activity. Amer. J. Anat. 90, 339–377 (1952).
Moss, D. W., Eaton, R. H., Smith, J. K., Whiteby, L. G.: Alterations in the electrophoretic mobility of alkaline phosphatase after treatment with neuraminidase. Biochem. J. 98, 320–330 (1966).
Mowry, R. W.: Alcian blue technics for histochemical study of acidic carbohydrates. J. Histochem. Cytochem. 4, 407 (1956).
—: The special value of methods that colour with both acidic and vicinal hydroxyl groups in the histochemical study of mucins. With revised directions for the colloidal iron stain, the use of alcian blue 8 GX and their combinations with the periodic acid Schiff reaction. Ann. N. Y. Acad. Sci. 106, 402–423 (1963).
— Winkler, C. H.: The coloration of acidic carbohydrates of bacteria and fungi in tissue sections with special reference to capsules of Cryptococcus neoformans, Pneumococcus and Staphylococcus. Amer. J. Path. 32, 628–629 (1956).
Papkoff, H.: Glycoproteins with biological activity. In: Glycoproteins their composition, structure and function, ed. by Gottschalk A., p. 532–557. New York: Elsevier Publishing Co. 1966.
Plummer, T. H., Hirs, C. H. W.: The structure of bovine pancreatic ribonuclease B: Isolation of glycopeptide. J. biol. Chem. 239, 2530–2543 (1964).
Robinson, J. C., Pearce, J. E.: Differential action of neuraminidase on human serum alkaline phosphatases. Nature (Lond.) 204, 472–473 (1964).
Schultze, H. E.: Clinically interesting polysaccharide linked substances. Scand. J. clin Lab. Invest. 10, 9–13 (1958).
Scott, J. E., Dorling, J.: Differential staining of acid glycosaminoglycans (mucopolysaccharides) by alcian blue in salt solutions. Histochemie 5, 221–223 (1965).
— Quintarelli, G.: Differential staining of acid glycosaminoglycans by alcian blue in salt solutions. Biochem. J. 90, 4P-5P (1964).
Spicer, S. S.: A correlative study of the histochemical properties of rodent acid mucopolysaccharides. J. Histochem, Cytochem. 8, 18–35 (1960).
- Horn, R. G., Leppi, T. J.: Histochemistry of connective tissue mucopolysaccharides. In: The connective tissue. Int. Acad. Pathol. Monograph No 7, p. 251–303 (1967).
— Lillie, R. D.: Saponification as a means of selective reversing the methylation blockade of tissue basophilia. J. Histochem. Cytochem. 7, 123–125 (1959).
— Meyer, D. B.: Histochemical differentiation of acid mucopolysaccharides by means of combined aldehyde fuchsin-alcian blue staining. Amer. J. clin. Path. 33, 453–460 (1960).
— Stoward, P. J.: Suggestions for a histochemical terminology of carbohydrate-rich tissue components. J. Histochem. Cytochem. 13, 599–603 (1965).
— Warren, L.: The histochemistry of sialic acid containing mucoproteins. J. Histochem Cytochem. 8, 135–137 (1960).
Svensmark, O.: Effects of sialidase on the electrophoretic properties of human serum cholinesterase. Dan. med. Bull. 8, 28–29 (1961).
— Kristensen, P.: Electrophoretic mobility of sialidase-treated human serum cholinesterase. Dan. med. Bull. 9, 16–17 (1962).
Watanabe, K., Fishman, W. H.: Application of the stereospecific inhibitor L-phenylalanine to the enzymorphology of intestinal alkaline phosphatase. J. Histochem. Cytochem. 12, 252–260 (1964).
Wislocki, G. B., Bunting, H., Dempsey, E. W.: Metachromasia in mammalian tissues and its relationship to mucopolysaccharides. Amer. J. Anat. 81, 1–37 (1947).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Varute, A.T., Patil, V.A. Histochemical analysis of molluscan stomach and intestinal alkaline phosphatase: A sialoglycoprotein. Histochemie 25, 77–90 (1971). https://doi.org/10.1007/BF00303949
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00303949