Abstract
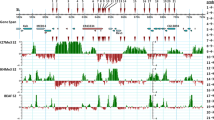
The distribution patterns of chromosomal proteins from Drosophila can be observed by immunofluorescent staining of the polytene chromosomes from larval salivary glands. We have purified a non-histone chromosomal protein of Mr=69 000 molecular weight which has a high affinity for DNA with little sequence specificity. Immunofluorescent staining indicates that this protein is preferentially associated with the inactive portions of the genome, including the centric heterochromatin and the condensed bands within the euchromatic arms of the chromosomes. Observation of both the heat shock loci 87A and 87C and the developmentally regulated loci 74EF and 75B shows an inverse correlation between immunofluorescent staining for the Mr=69 000 protein and for RNA polymerase. The presence of this protein appears to be correlated with the packaging of the chromatin in an inactive form.
Similar content being viewed by others
References
Alfageme CR, Rudkin JT, Cohen LH (1976) Locations of chromosomal proteins in polytene chromosomes. Proc Natl Acad Sci USA 73:2038–2042
Alfageme CR, Rudkin JT, Cohen LH (1980) Isolation, properties and cellular distribution of D1, a chromosomal protein of Drosophila. Chromosoma 78:1–31
Ashburner M (1972) Puffing patterns in Drosophila melanogaster and related species. In: Beermann W (ed) Developmental studies on giant chromosomes. Springer, New York, pp 101–151
Bonner JJ, Pardue ML (1976) The effect of heat shock on RNA synthesis in Drosophila tissues. Cell 8:43–50
Cartwright IL, Elgin SCR (1986) Nucleosomal instability and induction of new upstream protein-DNA associations accompany activation of four small heat shock protein genes in Drosophila melanogaster. Mol Cell Biol 6:779–791
Cheney CM, Shearn A (1983) Developmental regulation of Drosophila imaginal disc proteins: Synthesis of a heat shock protein under non-heat-shock conditions. Dev Biol 95:325–330
Christensen ME, LeStourgeon WM, Jamrich M, Howard GC, Serunian LA, Silver LM, Elgin SCR (1981) Distribution studies on polytene chromosomes using antibodies directed against hnRNP. J Cell Biol 90:18–24
Cohen LH, Gotchel DV (1971) Histones of polytene and non polytene nuclei of Drosophila melanogaster. J Biol Chem 246:1841–1848
Elgin SCR, Boyd JB (1975) The proteins of polytene chromosomes of Drosophila hydei. Chromosoma 51:135–145
Elgin SCR, Weintraub H (1975) Chromosomal proteins and chromatin structure. Annu Rev Biochem 44:725–774
Elgin SCR, Serunian LA, Silver LM (1978) Distribution patterns of Drosophila nonhistone chromosomal proteins. Cold Spring Harbor Symp Quant Biol 42:839–850
Fleischmann G, Pflugfelder G, Steiner EK, Javaherian K, Howard GC, Wang JC, Elgin SCR (1984) Drosophila DNA topoisomerase I is associated with transcriptionally active regions of the genome. Proc Natl Acad Sci USA 81:6958–6962.
Garel A, Axel R (1976) Selective digestion of transcriptionally active ovalbumin genes from oviduct nuclei. Proc Natl Acad Sci USA 73:3966–3970
Gilmour DS, Lis JT (1985) In vivo interactions of RNA polymerase with genes of Drosophila melanogaster. Mol Cell Biol 5:2009–2018
Heller S, Shelton ER, Dietrich V, Elgin SCR, Brutlag DL (1986) Multiple forms and cellular localization of Drosophila DNA topoisomerase II. J Biol Chem 261:8063–8069
Hewisch D, Burgoyne L (1973) Chromatin sub-structure. The digestion of chromatin DNA at regularly spaced sites by a nuclear deoxyribonuclease. Biochem Biophys Res Commun 52:504–510
Hill RJ, Mott MR, Burnett EJ, Abmayr SM Lowenhaupt K, Elgin SCR (1982) Nucleosome repeat structure is present in native salivary chromosomes of Drosophila melanogaster. J Cell Biol 95:262–266
Hovemann B, Galler R (1982) Vitellogenin in Drosophila melanogaster: a comparison of the YPI and YPII genes and their transcription products. Nucleic Acids Res 10:2261–2274
Ireland RC, Berger E, Sirotkin K, Yund MA, Osterbur D, Fristrom J (1982) Ecdysterone induces the transcription of four heat shock genes in Drosophila S3 cells and imaginal discs. Dev Biol 93:498–507
James TC, Elgin SCR (1986) Identification of a nonhistone chromosomal protein associated with heterochromatin in Drosophila melanogaster and its gene. Mol Cell Biol 6:3862–3872
Jamrich M, Greenleaf AL, Bautz EKF (1977a) Localization of RNA polymerase in polytene chromosomes of Drosophila melanogaster. Proc Natl Acad Sci USA 74:2079–2083
Jamrich M, Haars R, Wulf E, Bautz EKF (1977b) Correlation of RNA polymerase B and transcriptional activity in the chromosomes of Drosophila melanogaster. Chromosoma 64:319–326
Javaherian K, Tse Y-C, Vega J (1982) Drosophila topoisomerase I: isolation, purification and characterisation. Nucleic Acids Res 10:6945–6955
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Mayfield JE, Serunian LA, Silver LM, Elgin SCR (1978) A protein released by DNase I digestion of Drosophila nuclei is preferentially associated with puffs. Cell 14:539–544
McKenzie SL, Henikoff S, Meselson M (1975) Localization of RNA from heat-induced polysomes at puff sites in Drosophila melanogaster. Proc Natl Acad Sci USA 72:1117–1121
Peterson JL, McConkey EH (1976) Non-histone proteins from HeLa cells. J Biol Chem 251:548–554
Plagens U, Greenleaf AL, Bautz EKF (1976) Distribution of RNA polymerase on Drosophila polytene chromosomes as studied by indirect immunofluorescence. Chromosoma 59:157–165
Riggs AD, Suzuki H, Bourgeois S (1970) lac repressor-operator interactions I. equilibrium studies. J Mol Biol 48:67–83
Ritossa F (1962) A new puffing pattern induced by temperature shock and DNP in Drosophila. Experientia 18:571–573
Saumweber H, Symmons P, Kabisch R, Will H, Bonhoeffer F (1980) Monoclonal antibodies against chromosomal proteins of Drosophila melanogaster. Establishment of antibody producing cell lines and partial characterisation of corresponding antigens. Chromosoma 80:255–288
Silver LM, Elgin SCR (1976) A method for determination of the in situ distribution of chromosomal proteins. Proc Natl Acad Sci USA 73:423–427
Silver LM, Elgin SCR (1977) Distribution patterns of three subfractions of Drosophila nonhistone chromosomal proteins: possible correlations with gene activity. Cell 11:971–983
Sirotkin K, Davidson N (1982) Developmentally regulated transcription from Drosophila melanogaster chromosomal site 67B. Dev Biol 89:196–210
Spradling A, Penman S, Pardue ML (1975) Analysis of Drosophila mRNA by in situ hybridization: sequences transcribed in normal and heat shocked cultured cells. Cell 4:395–404
Tissieres A, Mitchell HK, Tracy UM (1974) Protein synthesis in salivary glands of Drosophila melanogaster: relation to chromosome puffs. J Mol Biol 84:389–398
Tjian R, Stinchcomb D, Losick R (1974) Antibody directed against Bacillus subtilis σ factor purified by sodium dodecyl sulfate slab gel electrophoresis. J Biol Chem 250:8824–8828
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Weeks JR, Coulter DE, Greenleaf AL (1982) Immunological studies of RNA polymerase II using antibodies to subunits of Drosophila and wheat germ enzyme. J Biol Chem 257:5884–5891
Weintraub H, Groudine M (1976) Chromosome subunits in active genes have an altered conformation. Science 193:848–856
Woodcock CLF, Safer JP, Stanchfield JES (1976) Structural repeating units in chromatin. Exp Cell Res 97:101–110
Wu C, Bingham PM, Livak KJ, Holmgren R, Elgin SCR (1979a) The chromatin structure of specific genes. I. Evidence for higher order domains of defined DNA sequences. Cell 16:797–806
Wu C, Wong Y-C, Elgin SCR (1979b) The chromatin structure of specific genes: II. Disruption of chromatin structure during gene activity. Cell 16:807–814
Zimmerman JL, Petri W, Meselson MS (1983) Developmental regulation of Drosophila imaginai disc proteins: Synthesis of a heat shock protein under non-heat-shock conditions. Cell 32:1161–1169
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fleischmann, G., Filipski, R. & Elgin, S.C.R. Isolation and distribution of a Drosophila protein preferentially associated with inactive regions of the genome. Chromosoma 96, 83–90 (1987). https://doi.org/10.1007/BF00285889
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00285889