Summary
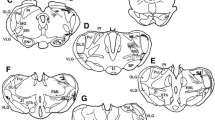
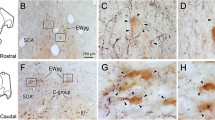
Injection of horseradish peroxidase (HRP) into the dorsal lateral geniculate nucleus (LGN) of the rabbit gave rise to retrograde labeling of neurones in the caudal part of the thalamic reticular nucleus. Electrophysiological observations demonstrated that these neurones met all criteria for interneurones in the recurrent inhibitory circuit of the geniculo-cortical pathway. They responded to stimulation of the visual cortex (Cx) or the optic chiasm (OX) with a burst of repetitive discharges, in agreement with the long-lasting IPSP from Cx or OX in relay cells of LGN. Results of collision test showed that the reticular neurones received excitatory input via axonal collaterals of relay cells. The latency of their response to stimulation of Cx or OX is about 1.8 ms shorter than that of the corresponding IPSP in the relay cells. Stimulation of LGN evoked an antidromic spike in reticular neurones with a latency of bout 1.1 ms, indicating a monosynaptic projection from the latter to the relay cells. All evidence indicates that interneurones in the recurrent inhibitory circuit are most likely located in the caudal part of the thalamic reticular nucleus of the rabbit.
Similar content being viewed by others
References
Ahlsén G, Lindström S, Sybirska E (1978) Subcortical axon collaterals of principal cells in the lateral geniculate body of the cat. Brain Res 156: 106–109
Ahlsén G, Lindström S (1978a) Axonal branching of functionally identified neurones in the lateral geniculate body of the cat. Neurosci Lett Suppl 1: 156
Ahlsén G, Lindström S (1978b) Projection of perigeniculate neurones to the lateral geniculate body in the cat. Neurosci Lett Suppl 1: 367
Ahlsén G, Lindström S (1982) Excitation of perigeniculate neurones via axon collaterals of principal cells. Brain Res 236: 477–481
Ahlsén G, Lindström S, Lo F-S (1982) Functional distinction of perigeniculate and thalamic reticular neurons in the cat. Exp Brain Res 46: 118–126
Ahlsén G, Lindström S, Lo F-S (1983) Excitation of perigeniculate neurones from X and Y principal cells in the lateral geniculate nucleus of the cat. Acta Physiol Scand 118: 445–448
Ahlsén G, Lindström S (1983) Corticofugal projection to perigeniculate neurones in the cat. Acta Physiol Scand 118: 181–184
Boyapati J, Henry G (1984) Corticofugal axons in the lateral geniculate nucleus of the cat. Exp Brain Res 53: 335–340
Curtis DR, Tebecis AK (1972) Bicuculline and thalamic inhibition. Exp Brain Res 16: 210–218
Dubin MW, Cleland BG (1977) Organization of visual inputs to interneurons of lateral geniculate nucleus of the cat. J Neurophysiol 40: 410–427
Fuster JM, Creutzfeldt OD, Straschill M (1965) Intracellular recording of neuronal activity in the visual system. Z Vergl Physiol 49: 605–622
Giolli RA, Towns LC, Takahashi TT, Karamanlidis AN, Williams DD (1978) An autoradiographic study of the projection of visual cortical area 1 to the thalamus, pretectum and superior colliculus of the rabbit. J Comp Neurol 180: 743–752
Houser CR, Vaughn JE, Barker R, Roberts E (1980) GABA neurons are the major cell type of the nucleus reticularis thalami. Brain Res 200: 341–345
Hughes A (1971) Topographical relationships between the anatomy and physiology of the rabbit visual system. Docum Ophthal 30: 33–159
Ide LS (1982) The fine structure of the perigeniculate nucleus in the cat. J Comp Neurol 210: 317–334
Jones EG (1975) Some aspects of the organization of the thalamic reticular complex. J Comp Neurol 162: 285–308
Lindström S (1982) Synaptic organization of inhibitory pathways to principal cells in the lateral geniculate nucleus of the cat. Brain Res 234: 447–453
Lo F-S (1981) Synaptic organization of the lateral geniculate nucleus of the rabbit: lack of feed-forward inhibition. Brain Res 221: 387–392
Lo F-S (1983a) Both fast and slow relay cells in lateral geniculate nucleus of rabbits receive recurrent inhibition. Brain Res 271: 335–338
Lo F-S (1983b) Lack of binocular inhibition in monocular segment of lateral geniculate nucleus of rabbits. Brain Res 271: 339–341
Lo F-S (1983c) Further study of neuronal circuit in lateral geniculate nucleus of rabbits. Neurosci Lett Suppl 12: S36
Lo F-S (1985) Responses of the rabbit lateral geniculate neurones to electrical stimulation of the visual cortex. Chinese J Physiol Sci 1: 129–136
Magnin M, Putkonen PTS (1978) A new vestibular thalamic area: electrophysiological study of the thalamic reticular nucleus and of the ventral lateral geniculate complex of the cat. Exp Brain Res 32: 91–104
Mesulam M-M (1981) Enzyme histochemistry of horseradish for tracing neural connections with the light microscope. In: Johnson JE Jr (ed) Current trends in morphological techniques. CRC Press Inc, Boca Raton, Florida, Vol I, pp 1–54
Montero VM, Guillery RW, Woolsey CN (1977) Retinotopic organization within the thalamic reticular nucleus demonstrated by a label autoradiographic technique. Brain Res 138: 407–421
Montero VM, Scott GL (1981) Synaptic terminals in dorsal lateral geniculate nucleus from neurons of the thalamic reticular nucleus. A light and electron microscope autoradiographic study. Neuroscience 6: 2561–2577
Montero VM, Singer W (1984) Ultrastructure and synaptic relations of neural elements containing glutamic acid decarboxylase (GAD) in the perigeniculate nucleus of the cat. A light and electron microscopic immunocytochemical study. Exp Brain Res 56: 115–125
Morgan R, Sillito AM, Wolstencroft JH (1974) A pharmacological investigation of inhibition in the lateral geniculate nucleus. J Physiol (London) 246: 93P
Ohara PT, Sefton AJ, Lieberman AR (1980) Mode of termination of afferents from the thalamic reticular nucleus in the dorsal lateral geniculate nucleus of the rat. Brain Res 197: 503–506
Ohara PT, Lieberman AR (1981) Thalamic reticular nucleus: anatomical evidence that cortico-reticular axons establish monosynaptic contact with reticulo-geniculate projection cells. Brain Res 207: 153–156
Sawyer CH, Everett JW, Green JD (1954) The rabbit diencephalon in stereotaxic coordinates. J Comp Neurol 101: 801–824
Sugitani M (1979) Electrophysiological and sensory properties of the thalamic reticular neurones related to somatic sensation in rats. J Physiol (London) 290: 79–95
Sumitomo I, Nakamura M, Iwama K (1976a) Location and function of the so-called interneurons of rat lateral geniculate body. Exp Neurol 51: 110–123
Sumitomo I, Iwama K, Nakamura M (1976b) Optic nerve innervation of so-called interneurons of the rat lateral geniculate body. Tohoku J Exp Med 119: 149–158
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lo, F.S., Xie, G.Y. Location of interneurones in the recurrent inhibitory circuit of the rabbit lateral geniculate nucleus. Exp Brain Res 66, 83–89 (1987). https://doi.org/10.1007/BF00236204
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00236204