Summary
The membrane electric effects of N,N′-dicyclohexylcarbodiimide (DCCD) and vanadate were studied in murine erythroleukemia cells (MELC), comparing the patch-clamp technique and the accumulation ratio (AR exp) of [3H]-tetraphenylphosphonium (TPP+). Electrophysiological measurements showed that both these inhibitors produce, at micromolar concentrations, a 20–30 mV hyperpolarization of resting potential (Δψ p ) of MELC, which is abolished when the electrochemical equilibrium potential of K+ (E K) is brought close to zero.
DCCD and vanadate turned out to have distinct targets on the plasma membrane of MELC (an H+ pump and the Na+,K+-ATPase, respectively).
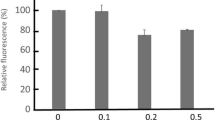
Measurements of AR exp showed that: (i) patch-clamp measurements of Δψ p were equivalent to those based on ARexp of antimycin-pretreated cells (AR ANT); (ii) DCCD produced a strong increase in AR ANT, that was antagonized by carbonyl cyanide p-trifluoromethoxyphenyl-hydrazone (FCCP) and diethylstilbestrol (DES); (iii) vanadate determined a marked increase in AR ANT that was insensitive to FCCP, but antagonized by ouabain; (iv) incubation in high K+ medium (HK) brought ARANT to 1.0 in the controls, but did not lower this ratio below 3.0 in the presence of DCCD or vanadate; (v) the total amount of TPP+ taken up by the cells was in any case water extractable by a freezing and thawing procedure.
On the whole, our data indicate that DCCD and vanadate hyperpolarize the MELC by increasing the K+ conductance and, at the same time, enhance the TPP+ binding, probably by changing the electrostatic potential profile of the plasma membrane. These effects seem to involve functional modifications of the target pumps, apparently related to the ion-occluding state of these enzymes.
We gratefully acknowledge the revision of the English by Dr. R. Manning of the Department of Biological Sciences, University of Durham, Durham, UK.
A. Arcangeli was supported by a fellowship from the Associazione Italiana contro le Leucemie (AIL); A. Becchetti and M.R. Del Bene were supported by fellowships from the Italian Association for Cancer Research (AIRC).
This work was supported by grants from the AIRC, from the Consiglio Nazionale delle Ricerche (CNR) (Special project “Ion channels”) and from the Ministero Pubblica Istruzione.
Similar content being viewed by others
References
Al-Awqati, Q. 1986. Proton-translocating ATPases. Annu. Rev. Cell Biol. 2:179–199
Amzel, L.M., Pedersen, L. 1983. Proton ATPases: Structure and mechanism. Annu. Rev. Biochem. 52:801–824
Andersen, O.S., Feldberg, S., Nakadomari, H., Levy, S., McLaughlin, S. 1978. Electrostatic interactions among hydrophobic ions in lipid bilayer membranes. Biophys. J. 21:35–70
Arcangeli, A., Del Bene, M.R., Olivotto, M., Wanke, E. 1989b. Ion channels in cancer cells. In: Membrane Technology. Vol. 64, pp. 65–79, Raven, New York
Arcangeli, A., Del Bene, M.R., Poli, R., Ricupero, L., Olivotto, M. 1989a Mutual contact of murine erythroleukemia cells activates depolarizing cation channels, whereas contact with extracellular substrata activates depolarizing Ca2+-dependent K− channels. J. Cell. Physiol. 139:1–8
Arcangeli, A., Olivotto, M. 1986. Plasma membrane potential of murine erythroleukemia cells: Approach to measurement and evidence for cell density-dependence. J. Cell. Physiol. 127:17–27
Arcangeli, A., Ricupero, L., Olivotto, M. 1987a Commitment to differentiation of murine erythroleukemia cells involves a modulated plasma membrane depolarization through Ca2+-activated K+ channels. J. Cell. Physiol. 132:387–400
Arcangeli, A., Wanke, E., Olivotto, M., Camagni, S., Ferroni, A. 1987b Three types of ion channels are present on the plasma membrane of Friend erythroleukemia cells. Biochem. Biophys. Res. Commun. 146:1450–1457
Ashcroft, F.M., Harrison, D.E., Ashcroft, S.J.H. 1984. Glucose induces closure of single potassium channels in isolated rat panceratic β-cells. Nature 312:446–448
Azzone, G.F., Pietrobon, D., Zoratti, M. 1984. Determination of the proton electrochemical gradient across biological membranes. Curr. Top. Bioenerg. 13:1–77
Bakker, E.P. 1982. Membrane potential in a potassium transportnegative mutant of Escherichia coli K-12. The distribution of rubidium in the presence of valinomycin indicates a higher potential than that of the tetraphenylphosphonium cation. Biochim. Biophys. Acta 681:474–483
Baroni, M., Arcangeli, A., Di Pietro, L., Alberghina, L. 1988. Plasma membrane potential of budding yeast is modulated by glucose and by inhibitors of H+-ATPase and of K+ channels. Yeasts 4:S34
Beaugé, L. 1988. Inhibition of translocation reaction by Vanadate. Methods Enzymol. 156:251–267
Benos, D.J. 1982. Amiloride: A molecular probe of sodium transport in tissues and cells. Am. J. Physiol. 242:C131-C145
Cantley, L.C., Resh, M.D., Guidotti, G. 1978. Vanadate inhibits the red cell (Na+,K+)-ATPase from the cytoplasmic side. Nature 272:552–554
De La Peña, P., Barros, F., Gascon, S., Ramos, S., Lazo P.S. 1982. The electrochemical proton gradient of Saccharomyces. Eur. J. Biochem. 123:447–453
Felber, S.M., Brand, M.D. 1982. Factors determining the plasmamembrane potential of human lymphocytes. Biochem. J. 204:577–585
Flewelling, R.F., Hubbell, W.L. 1986a Hydrophobic ion interaction with membranes. Biophys. J. 49:531–540
Flewelling, R.F., Hubbell, W.L. 1986b The membrane dipole potential in a total membrane potential model. Biophys. J. 49:541–552
Forbush, B., III. 1987. Rapid release of 42K and 86Rb from an occluded state of the Na,K-pump in the presence of ATP or ADP. J. Biol. Chem. 262:11104–11115
Futai, M., Noumi, T., Maeda, M. 1989. ATP synthase (H+-ATPase): Results by combined biochemical and molecular biological approaches. Annu. Rev. Biochem. 58:111–136
Glynn, I.M., Karlish, S.J.D. 1975. The sodium pump. Annu. Rev. Physiol. 37:13–55
Glynn, I.M., Richards, D.E. 1982. Occlusion of rubidium ions by the sodium-potassium pump: Its implications for the mechanism of potassium transport. J. Physiol. 330:17–43
Goffeau, A., Slayman, C.W. 1981. The proton-translocating ATPase of fungal plasma membrane. Biochim. Biophys. Acta 636:197–223
Hamill, O.P. 1983. Potassium and chloride channels in red blood cells. In: Single-channel Recording. B. Sackmann and E. Neher, editors. pp. 451–471, Plenum, New York
Hamill, O.P., Marty, A., Neher, E., Sakmann, B., Sigworth, F. 1981. Improved patch-clamp techniques for high resolution current recording from cells and cell-free membrane patches. Pfluegers Arch. 391:85–100
Hladky, S.B. 1979. The carrier mechanism. Curr. Top. Membr. Transp. 12:53–164
Hootman, S.R., Ernst, S.A. 1988. Estimation of Na+,K+-pump numbers and turnover in intact cells with [3H]ouabain. Methods Enzymol. 156:213–229
Hoppe, J., Sebald, W. 1984. The proton conducting F0-part of bacterial ATP synthases. Biochim. Biophys. Acta 768:1–27
Jordan, P.C. 1983. Electrostatic modeling of ion pores.II. Effects attributable to the membrane dipole potential. Biophys. J. 41:189–195
Ketterer, B., Neumcke, B., Läuger, P. 1971. Transport mechanism of hydrophobic ions through lipid bilayer membranes. J. Membrane Biol. 5:225–245
Kleijn, W.B., Bruner, L.J., Midland, M.M., Wisniewski, S. 1983. Hydrophobic ion probe studies of membrane dipole potentials. Biochim. Biophys. Acta 727:357–366
Lichtshtein, D., Mullikin-Kilpatrick, D., Blume, A.J. 1982. Hyperpolarization of neuroblastoma-glioma hybrid NG 108–15 by vanadium ions. Proc. Natl. Acad. Sci. USA 79:4202–4206
McEnery, M.W., Hullihen, J., Pedersen, P.L. 1989. F0 “proton channel” of rat liver mitochondria. J. Biol. Chem. 264:12029–12036
McEnery, M.W., Pedersen, P.L. 1986. Diethylstilbestrol. A novel F0-directed probe of the mitochondrial proton ATP-ase. J. Biol. Chem. 261:1745–1752
McLaughlin, S. 1977. Electrostatic potentials at membrane-solution interfaces. Curr. Top. Membr. Transp. 25:71–144
McLaughlin, S. 1989. The electrostatic properties of membranes. Annu. Rev. Biophys. Chem. 18:113–136
McLaughlin, S., Whitaker, M. 1988. Cations that alter surface potentials of lipid bilayers increase the calcium requirement for exocytosis in sea urchin eggs. J. Physiol. 396:189–204
Mitchell, P. 1961. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 191:144–148
Neumcke, B., Läuger, P. 1969. Nonlinear electrical effects in lipid bilayer membranes. II. Integration of the generalized Nernst-Planck equations. Biophys. J. 9:1160–1170
Nicholls, D.G. 1982. Bioenergetics. An introduction to the Chemiosmotic Theory. Academic, London
Pedersen, P.L., Carafoli, E. 1987a Ion motive ATPases. I. Ubiquity, properties and significance to cell function. Trends Biochem. Sci. 12:146–150
Pedersen, P.L., Carafoli, E. 1987b. Ion motive ATPases. II. Energy coupling and work output. Trends Biochem. Sci. 12:186–189
Penefsky, H.S. 1979. Mitochondrial ATPase. Adv. Enzymol. 49:223–280
Pogolotti, A.L., Santi, D.V. 1982. High pressure liquid chromatography-ultraviolet analysis of intracellular nucleotides. Anal. Biochem. 126:335–345
Post, R.L., Hegyvary, C., Kume, S. 1972. Activation by adenosin triphosphate in the phosphorylation kinetics of sodium and potassium ion transport adenosine triphosphatase. J. Biol. Chem. 247:6530–6540
Ramirez, J.A., Vacata, V., McCusker, J.H., Haber, J.E., Mortimer, R.K., Owen, W.G., Lecar, H. 1989. ATP-sensitive K+ channels in a plasma membrane H+-ATPase mutant of the yeast Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 86:7866–7870
Ritchie, R.J. 1984. A critical assessment of the use of lipophilic cations as membrane potential probes. Prog. Biophys. Mol. Biol. 43:1–32
Rottenberg, H. 1979. The measurement of membrane potential and pH in cells, organelles and vesicles. Methods Enzymol. 55:547–569
Skou, J.C. 1988. Overview: The Na,K-pump. Methods Enzymol. 156:1–25
Swallow, C.J., Grinstein, S., Rotstein, O.D. 1988. Cytoplasmic pH regulation in macrophages by an ATP-dependent and N,N′-dicyclohexilcarbodiimide-sensitive mechanism. J. Biol. Chem. 263:19558–19563
Wallick, E.J., Schwartz, A. 1988. Saturation of cardiac glycosides with Na+,K+-ATPase. Methods Enzymol. 156:201–213
Author information
Authors and Affiliations
Additional information
The authors are indebted to Prof. A. Fonnesu, Chairman of the Institute of General Pathology of the University of Florence, for his support and advice. Thanks are also due to Prof. A. Ferroni, for the critical reading of the manuscript and to Prof. S. Capaccioli for his contribution to ATP determination. We are grateful to M. Cutrì for technical assistance.
Rights and permissions
About this article
Cite this article
Arcangeli, A., Del Bene, M.R., Becchetti, A. et al. Effects of inhibitors of ion-motive ATPases on the plasma membrane potential of murine erythroleukemia cells. J. Membarin Biol. 126, 123–136 (1992). https://doi.org/10.1007/BF00231911
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00231911