Summary
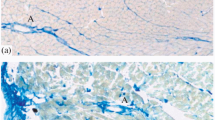
Previous studies have indicated that the asymmetric form of acetylcholinesterase (collagen-tailed) is localized in the basal lamina of the neuromuscular junction of skeletal muscle. The present study shows localization of the asymmetric acetylcholinesterase in the heart of the rat. Antiserum to 14+18 S acetylcholinesterase of the electric eel was raised in rabbits. The purified antibody did not react with collagen type I or laminin. Collagenase reduced the immunoreactivity of the enzyme with the purified antibody. Isolated cardiomyocytes and frozen sections of the heart were stained for acetylcholinesterase with the antibody. Diffuse immunofluorescence appeared over the surface of the cardiomyocytes. In the frozen sections, the immunofluorescence was most intense at the cell boundaries. These data suggest that collagenase-sensitive acetylcholinesterase in the heart is present in the myocytes and occurs in the vicinity of the basal lamina.
Similar content being viewed by others
Abbreviations
- AChE :
-
acetylcholinesterase
- BSA :
-
bovine serum albumin
- PBS :
-
phosphate-buffered saline
- DME :
-
Dulbecco's Modified Eagle Medium
References
Anglister L, Silman I (1978) Molecular structure of enlongated forms of electric eel acetylcholinesterase. J Mol Biol 125:293–311
Anglister L, McMahan UJ (1985) Basal lamina directs acetylcholinesterase accumulation at synaptic sites in regenerating muscle. J Cell Biol 101:735–743
Anglister L, Tarrab-Hazdai R, Fuchs S, Silman I (1978) Immunological cross-reactivity between electric eel acetylcholinesterase and rat tail tendon collagen. Eur J Biochem 94:25–29
Bon S, Vigny M, Massoulie J (1979) Asymmetric and globular forms of acetylcholinesterase in mammals and birds. Proc Natl Acad Sci USA 76:2546–2550
Brimijoin S, Rakonczay Z, Mintz K (1986) Immunochemistry of mammalian cholinesterases. Fed Proc 54:2960–2964
Buxton ILO, Brunton LL (1986) Compartmentation of hormone action. Adv Exp Med Biol 194:117–127
Couteaux R (1955) Localization of cholinesterases at neuromuscular junction. Int Rev Cytol 4:355–375
Eghbali M, Seifter S, Robinson TF, Blumenfeld OO (1987) Enzyme-antibody histochemistry: A method for detection of collagens collectively. Histochemistry 87:257–262
Engval E (1980) Enzyme immunoassay ELISA and EMIT. Methods Enzymol 70:419–439
Fonnum F (1969) Radiochemical microassays for the determination of choline acetyltransferase and acetylcholinesterase activities. Biochem J 115:465–472
Grassi J, Massoulie J, Timpl R (1983) Relationship of collagentailed acetylcholinesterase with basal lamina components: Absence of binding with laminin, fibronectin, and collagen typesIV and V and lack of reactivity with different anti-collagen sera. Eur J Biochem 133:31–38
Hall Z (1973) Multiple forms of acetylcholinesterase and their distribution in endplate and non-endplate regions of rat diaphragm muscle. J Neurobiol 4:343–361
Inestrosa NC, Silberstein L, Hall ZW (1982) Association of the synaptic form of acetylcholinesterase with extracellular matrix in cultured mouse muscle cells. Cell 29:71–79
Inestrosa NC, Roberts WL, Marshall TL, Rosenberry TL (1987) Acetylcholinesterase from bovine caudate nucleus is attached to membranes by a novel subunit distinct from those of acetylcholinesterases in other tissues. J Biol Chem 262:4441–4444
Kent MK, Cooper T (1974) The denervated heart; a model for studying antonomic control of the heart. New Engl J Med 291:1017–1021
Koelle GB (1963) Cytological distributions and physiological functions of cholinesterases. In: Handbuch der experimentellen Pharmakologie, XV, Springer, Berlin Heidelberg New York, 1987
Lyles JM, Silman I, DiGiamberardino L, Couraud JY, Barnard EA (1981) Comparison of the molecular forms of the cholinesterases in tissues of normal and dystrophic chickens. J Neurochem 38:1007–1021
Mark G, Chamley J, Burnstock G (1973) Interactions between autonomic nerves and smooth and cardiac muscle cells in tissue culture. Dev Biol 32:194–200
Marshall LM, Sanes JR, Memahan UJ (1977) Reinnervation of original synaptic sites on muscle fiber basement membrane after disruption of the muscle cells. Proc Natl Acad Sci USA 74:3073–3077
Massoulie J, Bon S (1982) The molecular forms of cholinesterase and acetylcholinesterase in vertebrates. Ann Rev Neurosci 5:57–106
Purves RD, Hill CE, Chamley J, Mark GE, Fry DM, Burnstock G (1974) Functional autonomic neuromuscular junctions in tissue culture. Pflügers Arch 350:1–7
Rosenberry TL, Richardson JM (1977) Structure of 18 S and 14 S acetylcholinesterase. Identification of collagen-like subunits that are linked by disulfide bonds to catalytic subunits. Biochemistry 16:3550–3558
Sandusky GE, White SL, Wightman KA (1986) Canine artioventricular node: Scanning electron microscopy and enzyme histochemistry. Am J Vet Res 47:304–308
Silman I, Futerman H (1987) Post-translational modification as a means of anchoring acetylcholinesterase to the cell surface. Biopolymers 26:S241-S253
Silman I, Lyles JM, Barnard EA (1978) Intrinsic forms of acetylcholinesterase in skeletal muscle. FEBS Lett 94:166–170
Silver A (1974) In: Neuberger A, Tatum EL (eds) The biology of cholinesterase in frontiers of biology. North-Holland, Amsterdam
Slavikova J, Ylk J, Hlavickova Y (1982) Acetylcholinesterase and butyrylcholinesterase activity in the atria of the heart of adult albino rats. Physiol Bohemoslov 31:407–417
Torigoe K, Nakamura T (1987) Fine structure of myomyous junctions in the mouse skeletal muscles. Tissue Cell 19:243–250
Vigny M, Koenig J, Rieger F (1976) The motor endplate specific form of acetylcholinesterase: Appearance during embryogenesis and reinnervation of rat muscle. J Neurochem 27:1347–1353
Vigny M, Martin GR, Grotendorst GR (1983) Interactions of asymmetric forms of acetylcholinesterase with basement membrane components. J Biol Chem 258:8794–8798
Wittenberg BA, Robinson TF (1981) Oxygen requirements, morphology, cell coat, and membrane permeability of calcium-tolerant myocytes from hearts of adult rats. Cell Tissue Res 216:231–251
Wittenberg BA, White RL, Ginzberg RD, Spray DC (1986) Effect of calcium on the dissociation of the mature rat heart into individual paired myocytes: Electrical properties of cell pairs. Circ Res 59:143–150
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eghbali, M., Silman, I., Robinson, T.F. et al. Visualization of collagenase-sensitive acetylcholinesterase in isolated cardiomyocytes and in heart tissue. Cell Tissue Res. 253, 281–286 (1988). https://doi.org/10.1007/BF00222282
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00222282