Summary
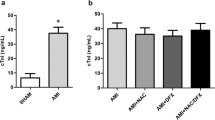
The glutathione redox pathway is an important antioxidant system in the myocardium. N-Acetylcysteine is a low molecular weight glutathione precursor that has been used clinically to replenish glutathione stores. The present study was aimed at evaluating the protective effect of N-acetylcysteine on myocardial damage resulting from permanent coronary occlusion (without reperfusion) in anaesthetized dogs. N-Acetylcysteine (150 mg kg−1 i.v.) administered 2 min before occlusion rerduced infarct size in dogs subjected to 24 h ischemia. The infarct size as a percentage of the area at risk was 86.8 ± 3.6% (n = 11) in control (salinetreated) dogs and 68.2 ± 2.4% (n = 7; P < 0.05 vs control) in N-acetylcysteine-treated animals. Haemodynamic variables (heart rate, mean arterial pressure and ratepressure product) were similar in the control and the treated group. Regional myocardial blood flow was determined with radioactive microspheres in ischaemic and non-ischaemic zones before occlusion and 3 h post-ocelusion. N-Acetylcysteine did not influence the regional distribution of myocardial blood flow. The myocardial content of reduced glutathione was significantly (P < 0.05) decreased 3 h post-occlusion (0.53 ± 0.19 μmol/ g−1 ; n = 5) compared to either pre-occlusion values (0.94 ± 0.03 μmol/g−1; n = 8) or values 3 h post-ocelusion in sham-operated animals (0.93 ± 0.15 μmo1/g−1 ; n = 5). Depletion of myocardial glutathione 3 h post-ocelusion was not observed in dogs treated with N-acetylcysteine (0.87 ± 0.11 μmol/g−1; n = 5). Superoxide dismutase activity and malondialdehyde levels were determined in blood samples obtained from the coronary vein draining the ischaemic zone. Superoxide dismutase activity increased 10 min post-occlusion in control but not in N-acetylcysteine-treated dogs. Malondialdehyde levels were elevated in both groups after occlusion but this increase failed to reach statistical significance in the animals treated with N-acetylcysteine. This study demon strates that N-acetylcysteine treatment reduces myocardial damage after permanent coronary occlusion. The beneficial effect may be due to maintenance of myocardial glutathione and to protection against free-radicalmediated damage during the early phase of ischaemia.
Similar content being viewed by others
References
Becker LC, Ambrosio G (1987) Myocardial consequences of reperfusion. Prog Cardiovasc Dis 30:23–44
Bernard GR, Lucht WD, Niedermeyer ME, Snapper JR, Ogletree ML, Brigham KL (1984) Effect of N-acetylcysteine on pulmonary response to endotoxin in the awake sheep and upon in vitro granulocyte function. J Clin Invest 73:1772–1784
Bonanomi L, Gazzaniga A (1980) Toxicological, pharmacokinetic and metabolic studies onacetylcysteine. Eur J Respir Dis 61 [Suppl 111]:45–51
Brigelius R, Muckel C, Akerboom TPM, Sies H (1983) Identification and quantification of glutathione in hepatic protein mixed disulfides and its relationship to glutathione disulfide. Biochem Pharmacol 32:2529–2534
Burton KP, McCord JM, Ghai G (1984) Myocardial alterations due to free-radical generation. Am J Physiol 246:H776-H783
Curello S, Ceconi C, Bigoli C, Ferrari R, Albertini A, Guarnieri C (1985) Changes in the cardiac glutathione status after ischemia and reperfusion. Experientia 41:42–43
Davies MJ (1989) Direct detection of radical production in the ischaemic and reperfused myocardium: current status. Free Radic Res Commun 7:275–284
Domenech RJ, Hoffman JIE, Noble MIM, Saunders KB, Henson JR, Subijanto S (1969) Total and regional coronary blood flow measured by radioactive microspheres in conscious and anesthetized dogs. Circ Res 25:581–596
Estrela JM, Saez GT, Such L, Viña J (1983) The effect of cysteine and N-acetylcysteine on rat liver glutathione (GSH). Biochem Pharmacol 32:3483–3485
Fantone JC, Ward PA (1982) Role of oxygen-derived free radicals and metabolites in leukocyte-dependent inflammatory reactions. Am J Pathol 107:397–418
Ferrari R, Ceconi C, Curello C, Guarnieri C, Caldaresa CM, Albertini A, Visoli O (1985) Oxygen-mediated myocardial damage during ischaemia and reperfusion: Role of the cellular defenses agaist oxygen toxicity. J Mol Cell Cardiol 17:937–945
Forman MB, Puett DW, Cates CU, McCroskey DE, Beckman JK, Greene HL, Virmani R (1988) Glutathione redox pathway and reperfusion injury. Effect of N-acetylcysteine on infarct size and ventricular function. Circulation 78:202–213
Fridovich I (1983) Superoxide radical: An endogenous toxicant. Annu Rev Pharmacol Toxicol 23:239–257
Gaitonde MK (1967) A spectrophotometric method for the direct determination of cysteine in the presence of other naturally occurring aminoacids. Biochem J 104:627–633
Kolodgie FD, Dawson AK, Forman MB, Virmani R (1985) Effect of perfluorochemical (Fluosol-DA) on infarct morphology in dogs. Virchows Arch [B] 50:119–134
Lange LG, Sobel BE (1982) Pharmacological salvage of myocardium. Annu Rev Pharmacol Toxicol 22:115–143
Lie JT, Pairolero PC, Holley KE, Titus JL (1975) Macroscopic enzyme-mapping verification of large, homogeneous, experimental myocardial infarcts of predictable size and location in dogs. J Thorac Cardiovasc Surg 69:599–605
Lucchesi BR, Mullane KM (1986) Leukocytes and ischemia-induced myocardial injury. Annu Rev Pharmacol Toxicol 26:201–224
Maroko PR, Kjekshus JK, Sobel BE, Watanabe T, Covell JW, Ross J Jr, Braunwald E (1971) Factors influencing infarct size following experimental coronary artery occlusions. Circulation 43:67–82
McCluskey ER, Murphree S, Saffitz JE, Morrison AR, Needleman P (1985) Temporal changes in 12-HETE formation in two models of canine myocardial infarction. Prostaglandins 29:387–403
McCord JM, Fridovich I (1969) Superoxide dismutase: an enzymic function for enrithrocuprein. J Biol Chem 244:6049–6052
Mitsos SE, Fantone JC, Gallagher KP, Walden KM, Simpson PJ, Abrams GD, Schork MA, Lucchesi BR (1986) Canine myocardial reperfusion injury: protection by a free radical scavenger, N-2-mercaptopropionyl glycine. J Cardiovasc Pharmacol 8:978–988
Mullane KM, Read N, Salmon JA, Moncada S (1984) Role of leukocytes in acute myocardial infarction in anesthetized dogs: relationship to myocardial salvage by anti-inflammatory drugs. J Pharmacol Exp Ther 228:510–522
Rao PS, Cohen MV, Mueller HS (1983) Production of free radicals and lipid peroxides in early experimental myocardial ischemia. J Mol Cell Cardiol 15:713–716
Romero FJ, Montoro A, Alberola A, Gil F, Saez GT, Viña J, Such L, (1987) Myocardial glutathione alterations in acute coronary occlusion in the dog. Free Radic Res Commun 4:27–30
Singh A, Lee KJ, Lee CY, Goldfarb RD, Tsan MF (1989) Relation between myocardial glutathione content and extent of ischemiareperfusion injury. Circulation 80:1795–1804
Such L, Morcillo E, Chorro FJ, Montoro A, Alberola A, Aparicio F, Viña J (1986) Beneficial effects of N-acetylcysteine on acute myocardial infarction in open-chest dogs. Arch Farmacol Toxicol 12:37–40
Tribble DL, Aw TY, Jones DP (1987) The pathophysiological significance of lipid peroxidation in oxidative cell injury. Hepatology 7:377–386
Uchiyama M, Mihara M (1978) Determination of malondialdehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:279–286
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alberola, A., Such, L., Gill, F. et al. Protective effect of N-acetylcysteine on ischaemia-induced myocardial damage in canine heart. Naunyn-Schmiedeberg's Arch Pharmacol 343, 505–510 (1991). https://doi.org/10.1007/BF00169553
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00169553