Abstract
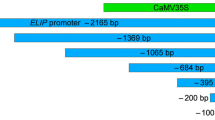
The chloroplastic Cu,Zn superoxide dismutase (SOD) has an important role in the defense against damage by oxygen radicals in the chloroplasts. Here, for the first time, we report on the isolation of a genomic DNA clone from tomato that contains all the coding sequence for the chloroplastic Cu,Zn SOD as well as a 442 bp DNA fragment upstream of the translational initiation site. The latter upstream sequence has a putative TATA box and a 285 bp promoter region, 5′ of the apparent transcriptional initiation and a 157 bp leader region. The coding sequence is composed of 8 exons that are interspaced by 7 introns; we termed this geneSODCp;Le:1. The 442 bp fragment was cloned into a pBI101 vector, upstream of theuidA (GUS) gene, via transcriptional fusion.Agrobacterium-mediated transformation resulted in transgenic tobacco plants.
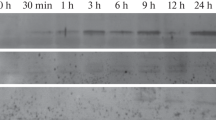
The progeny (after self-pollination) of 14 transformed plants, which expressed GUS above a threshold of 1 nmol/min per mg protein, were found to fall into two distinct groups. In the seedlings of 10 lines (group A) GUS expression was enhanced by exposure to light. In 4 lines of this group maintenance for 3 days in the dark eliminated GUS activity. The seedlings of group B expressed GUS regardless of the light/dark regime. In plants of group A, GUS expression was also developmentally regulated: high GUS activity in young leaves, low activity in mature leaves and no activity in the roots. The results suggest that this short chloroplastic Cu,Zn SOD promoter contains motifs for developmental (spatial) regulation as well as motifs responsive to light (or to oxygen radicals resulting from light-driven photosynthesis).
Similar content being viewed by others
References
Acevedo A, Scandalios JG: Differential expression of the catalase and superoxide dismutase genes in maize ear shoot tissues. Plant Cell Physiol 33: 1079–1088 (1992).
Asada K, Takahashi M: Production and scavenging of active oxygen in photosynthesis. In: Osmond DJ, Arntzen CJ (eds) Photoinhibition, pp. 227–287. Elsevier, Amsterdam (1987).
Ausubel FM: Current Protocols in Molecular Biology. Greene/Wiley, New York (1993).
Beauchamp CO, Fridovich I: Superoxide dismutase: improved assays and an assay applicable to acrylamide gel. Anal Biochem 44: 276–287 (1971).
Beyer W, Imlay J, Fridovich I: Superoxide dismutases. Prog Nucl Acids Res Mol Biol 40: 221–253 (1991).
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254 (1976).
Cavener DR, Ray SC: Eukaryotic start and stop translation sites, Nucl Acids Res 19: 3185–3192.
Galili S, Galili G, Feldman M. Chromosome location of genes for Rubisco small subunit and Rubisco-binding protein in common wheat. Theor Appl Genet 81: 98–104 (1991).
Gressel J, Galun E: Genetic controls of photooxidant tolerance. In: Foyer C, Mullineaux P (eds) Photooxidative Stresses on Plants: Causes and Amelioration. CRC Press, Boca Raton, FL, in press (1994).
Halliwell B, Gutteridge JMC: Free Radicals in Biology and Medicine, Clarendon Press, Oxford (1989).
Horsch RB, Fry JE, Hoffmann NL, Elchholtz D, Rogers SG, Fraley RT: A simple and general method for transfening genes into plants. Science 227: 1229–1231 (1985).
Jefferson RA, Kavanagh TA, Bevan MW: GUS fusion: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6: 3901–3907 (1987).
Kozak M: Point mutations define a sequence flanking the AUG initiation codon that modulates translation by eukaryotic ribosomes. Cell 44: 283–292 (1986).
Kitagawa Y, Tsunasawa S, Tanaka N, Katsube Y, Sakiyama F, Asada K: Amino acid sequence of copper, zinc-superoxide dismutase from spinach leaves. J Biochem 99: 1289–1298 (1986).
Kwaitowski J, Kaniuga Z: Isolation and characterization of cytosolic and chloroplast isozymes of Cu, Zn-superoxide dismutase from tomato leaves and their relationships to other Cu,Zn-superoxide dismutases. Biochim Biophys Acta 874: 99–115 (1986).
Laemmli AK: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685 (1970).
Li H-m, Washburn T, Chory J: Regulation of gene expression by light. Curr Opin Cell Biol 5: 455–460 (1993).
Lindsey K, Wei W, Clarke MC, McArdle HF, Rooke LM, Topping JF: Tagging genomic sequences that direct transgene expression by activation of a promoter trap in plants. Transgen Res 2: 33–47 (1993).
McCord JM, Fridovich I: Superoxide dismutase: an enzymatic function for erythrocuperin (hemocuperin). J Biol Chem 244: 6049–6055 (1969).
Perl-Treves R, Abu-Abied M, Magal N, Galun E, Zamir D. Genetic mapping of tomato cDNA clones encoding the chloroplastic and the cytosolic isozymes of super-oxide dismutase. Biochem Genet 28: 543–552 (1990).
Perl-Treves R, Galun E: The tomato Cu,Zn superoxide dismutase genes are developmentally regulated and respond to light and stress. Plant Mol Biol 17: 745–760 (1991).
Perl-Treves R, Nacmias B, Aviv D, Zeelon EP, Galun E: Isolation of two cDNA clones from tomato containing two different superoxide dismutase sequences. Plant Mol Biol 11: 609–623 (1988).
Rabinowitch HD, Fridovich I: Superoxide radicals, superoxide dismutases and oxygen toxicity in plants. Rev Photochem Photobiol 37: 679–690 (1983).
Sakamoto A, Okumura T, Ohsuga H, Tanaka K: Genomic structure of the gene for copper/zinc-superoxide dismutase in rice. FEBS Lett 301: 185–189 (1992).
Sambrook J, Fritsch EF, Maniatis T: Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY (1989).
Sanger F, Nicklen S, Coulson AR: DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74: 5463–5467 (1977).
Scioli JR, Zilinskas BA: Cloning and characterization of a cDNA encoding the chloroplastic copper/zinc-superoxide dismutase from pea. Proc Natl Acad Sci USA 85: 7661–7665 (1988).
Steffens GJ, Michelson AM, Otting F, Puget K, Strassburger W, Flohe L: Primary structure of Cu, Zn superoxide dismutase ofBrassica oleracea proves homology with corresponding enzymes of animals, fungi and prokaryotes. Biol Chem Hoppe-Seyler 367: 1007–1016 (1986).
Tepperman J, Katayama C, Dunsmuir P: Cloning and nucleotide sequence of a petunia gene encoding a chloroplast localized superoxide dismutase. Plant Mol Biol 11: 871–872 (1988).
Topping JF, Wei W, Lindsey K: Functional tagging of regulatory elements in the plant genome. Development 112: 1009–1019 (1991).
Tsang Ed WT, Bowler C, Herouart D, Camp WV, Villabroel R, Genetello C, Van Montagu M, Inze D: Differential regulation of superoxide dismutases in plants exposed to environmental stress. Plant Cell 3: 783–792 (1991).
Williamson JD, Scandalios JG: Plant antioxidant gene responses to fungal pathogens. Trends Microbiol 1: 239–245 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kardish, N., Magal, N., Aviv, D. et al. The tomato gene for the chloroplastic Cu,Zn superoxide dismutase: regulation of expression imposed in transgenic tobacco plants by a short promoter. Plant Mol Biol 25, 887–897 (1994). https://doi.org/10.1007/BF00028883
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00028883