Abstract
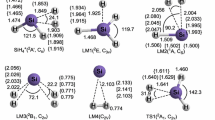
The mechanism of TiCl4 ammonolysis has been studied theoretically at ab initio Hartree-Fock, B3LYP, MP2 and CCSD(T)//B3LYP levels using effective core potentials for Ti and Cl and 6-31G* basis sets for N and H. TiCl4 and products of its ammonolysis form five- and six-coordinated complexes with ammonia, which intermediate substitution of Cl atoms by NH2 groups. Transition state energies for the subsequent steps of ammonolysis decrease with increasing number of NH2 groups bound to Ti. The energy of the transition state for the first step of ammonolysis is 19 kcal mol-1 above the energy of the reactants (TiCl4+NH3) and 8 kcal mol-1 above the products (TiCl3NH2 + HCl). The following steps have transition states energetically located below the products, indicating weak hydrogen bonded complex formation as intermediate between transition state and product. A thermodynamic estimation shows the last step of ammonolysis to be endothermic, while the first three steps are exothermic if the adduct formation energy is taken into account.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 11 November 1999/ Accepted: 18 January 2000/ Published: 28 April 2000
Rights and permissions
About this article
Cite this article
Siodmiak, M., Frenking, G. & Korkin, A. Theoretical ab initio Study of TiCl4 Ammonolysis: Gas Phase Reactions of TiN Chemical Vapor Deposition. J Mol Model 6, 413–424 (2000). https://doi.org/10.1007/PL00010740
Issue Date:
DOI: https://doi.org/10.1007/PL00010740