Summary
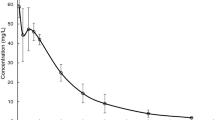
The pharmacokinetic disposition of prednisolone was studied following oral administration (10 mg) to eight patients with untreated coeliac disease of mild or moderate severity, seven coeliac patients on a strict gluten-free diet and ten normal subjects. No significant differences were shown in the peak prednisolone level, the time of the peak level, the area under the concentration versus time plot, the plasma half-life and the 24 h urinary recovery of prednisolone in the three subject groups. There was however considerable variability within each group. It is concluded that the presence of coeliac disease does not significantly alter the absorption or elimination of prednisolone.
Similar content being viewed by others
References
Heizer W.D., Smith T.W. and Goldfinger S.E. (1971): Absorption of Digoxin in patients with malabsorption syndromes. New. Eng. J. Med.,285, 257–259.
Parsons R.L., Hossack G. and Paddock G. (1975): The Absorption of antibiotics in adult patients with coeliac disease. J. Antimicrob. Chemother.,1, 39–50.
Parsons R.L., Kaye C.M., Raymond K., Trounce J.R. and Turner P. (1976): Absorption of propranolol and practolol in coeliac disease. Gut,17, 139–143.
Pickup M.E., Lowe J.R., Leatham P.A., Rhind V.M., Wright V. and Downie W.W. (1977): Dose Dependent Pharmacokinetics of Prednisolone. Europ. J. clin. Pharmacol.,12, 213–219.
Wagner J.G. (1967): Method of estimating relative absorption of a drug in a series of clinical studies in which blood levels are measured after single and/or multiple doses. J. Pharm. Sci.,56, 652–653.
Gibaldi M. and Perrier E. (1975): “Pharmacokinetics”, ed. J. Swarbrick, p. 145. Marcel Decker Inc., New York.
Morrison P.J., Bradbrook I.D. and Rogers H.J. (1977): Plasma prednisolone levels from enteric and non-enteric coated tablets estimated by an original technique. Br. J. clin. Pharmac.,4, 597–603.
Gibaldi M. and Perrier E. (1975): “Pharmacokinetics”, ed. J. Swarbrick, p. 153–166. Marcel Dekker Inc., New York.
Kozower M., Veatch L. and Kaplan M. (1974): Decreased Clearance of Prednisolone, a Factor in the Development of Corticosteroid Side Effects. J. Clin. Endocrinol. Metab.,38, 407–412.
Wilson C.G., Ssendagire R., May C.S. and Paterson J.W. (1975): Measurement of plasma prednisolone in man. Br. J. clin. Pharmac.,2, 321–325.
English J., Chakraborty J. and Marks V. (1974): A competitive protein binding method for plasma prednisolone assay. Ann. Clin. Biochem.,11, 11–14.
Meikle A.W., Weed J.A. and Tyler F.H. (1975): Kinetics and Interconversion of Prednisolone and Prednisone Studied with New Radioimmunoassays. J. clin. Endocrinol. Metab.,47, 717–721.
Pickup M.E. (1979): The Clinical Pharmacokinetics of Prednisone and Prednisolone. (A review) Clinical Pharmacokinetics,4, 111–128.
Leclercq R. and Copinschi G. (1974): Patterns of plasma levels of prednisolone after oral administration in man. J. Pharmacokin. Biopharm.,2, 175–187.
Davis M., Williams R., Chakraborty J., English J., Marks V., Ideo G. and Tempini S. (1978): Prednisone or prednisolone for the treatment of chronic active hepatitis? A comparison of plasma availability. Br. J. clin. Pharmac.,5, 501–505.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pickup, M.E., Farah, F., Lowe, J.R. et al. Prednisolone absorption in coeliac disease. European Journal of Drug Metabolism and Pharmacokinetics 4, 87–89 (1979). https://doi.org/10.1007/BF03189406
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03189406