Abstract
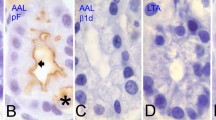
Biotechnology uses lectin genes to transfect into crop plants for protection against insects and nematodes. On the other hand, the information is limited on lectin-binding properties of cells in the gastrointestinal tract. Therefore, binding of a panel of FITC-labelled plant lectins to gastrointestinal cells of the rat was studied. In the stomach, cytoplasmic staining of parietal cells by PHA appeared to be due to glycoproteins attached to the tubulovesicles. PNA also stained the parietal cells, but only in the isthmus and neck regions, reacting with desialylated glycoproteins. WGA bound to the mucous neck cells with higher affinity than to the surface and foveolar mucous cells. The mucous cells were also stained by SNA-I, UEA-I and, less intensively, by LCA. Chief cells did not show detectable reaction with any of the applied lectins. Binding of PHA to gastric cells showed differences when compared with the results of in vivo studies. Small intestinal brush border was stained with UEA-I and SNA-I, the latter lectin also strongly stained the surface of small intestinal crypts. Both lectins reacted with the mucus of goblet cells. In the large intestine UEA-I and SNA-I stained the goblet cells at the base and upper part of the crypts, respectively. Accordingly, we provided evidences for the unique lectin-binding phenotype of the various segments of the gastrointestinal tract.
Similar content being viewed by others
References
Bardocz S, Grant G, Ewen SWB, et al: Reversible effect of phytohaemagglutinin on the growth and metabolism of rat gastrointestinal tract. Gut 37:353–360, 1995.
Broekaert WF, Nsimba-Lubaki M, Peeters B, Peumans WJ: A lectin from elder (Sambucus nigra L.) bark. Biochem J 221:163–169, 1984.
Callaghan JM, Toh B-H, Pettitt JM, et al: Poly-N-aceryllactosamine-specific tomato lectin interacts with gastric parietal cells. J Cell Science 95:563–576, 1990.
Ewen SWB, Pusztai A: Effects of diets containing genetically modified potatoes expressingGalanthus nivalis lectin on rat small intestine. Lancet 354:1353–4, 1999.
Fischer J: Alternative ultrastructural localization ofDolichos biflorus lectin binding sites in proton secreting parietal cells of mice. Biochemistry 87:1–4, 1989.
Goldkorn I, Gleeson PA andToh B-H: Gastric parietal cell antigens of 60–90 kDa, and 100–120 kDa associated with autoimmune gastritis and pernicious anaemia. Role of N-glycans in the structure and antigenicity of the 60–90 kDa component. J Biol Chem 264:18768–74, 1989.
Karam SM, Yao X, Forte JG: Functional heterogeneity of parietal cells along the pit-gland axis. Am J Physiol 272: G161-G171, 1997.
Pusztai, A: Plant Lectins. Cambridge University Press. 1991.
Pusztai A, Ewen SWB, Grant G et al: The relationship between survival and binding of plant lectins during small intestinal passage and their effectiveness as growth factors. Digestion 46: (suppl. 2) 308–316, 1990.
Schulte B andSpicer SS: Light microscopic histochemical detection of terminal galactose and N-acetylgalactosamine residues in rodent complex carbohydrates using a galactose oxidase-Schiff sequence and peanut lectin-horseradish peroxidase conjugate. J Histochem Cytochem 31:19–24, 1983.
Suzuki S, Tsuyama S andMurata F: Post-embedding staining of rat gastric mucous cells with lectins. Histochemistry 73:563–575, 1982.
Van Damme EJM, Barre A, Smeets K, et al: The bark ofRobinia pseudoacacia contains a complex mixture of lectins. Plant Physiol 107:833–843, 1995.
Van Damme EJM, Peumans WJ, Pusztai A, et al: Handbook of Plant Lectins. John Wiley & Sons, Chichester, 1998.
Wang Q, Lu-Gang Y, Campbell BJ, et al: Identification of intact lectin in peripheral venous blood. Lancet 352:1831–1832, 1998.
Zolotarev AS, Townsend RR, Stuart-Tilley A, et al: HCO3 −-dependent conformational change in gastric parietal cell AE2, a glycoprotein naturally lacking sialic acid. Am J Physiol 271:G311-G321, 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baintner, K., Jakab, G., Gyôri, Z. et al. Binding of FITC-labelled lectins to the gastrointestinal epithelium of the rat. Pathol. Oncol. Res. 6, 179–183 (2000). https://doi.org/10.1007/BF03032370
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03032370