Abstract
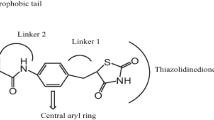
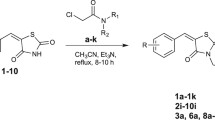
Benzothiazole derivatives of thiazolidinediones (TZD) were synthesized using a modified Mitsunobu reaction of 2-(benzothiazol-2-ylmethylamino)ethanol (2) with 5-(4-hydroxybenzyl)-3-triphenylmethylthiazolidine-2,4-dione and assayed for activity on peroxisome proliferator-activated receptor (PPAR) subtypes and inhibitory activity of NO production in lipopolysaccharide-activated macrophages. Most of the tested compounds were identified as potent PPARγ agonists, indicating their potential as drug candidates for diabetes.
Similar content being viewed by others
References
Cantello, B. C., Cawthorne, M. A., Cottam, G. P., Duff, P. T., Haigh, D., Hindley, R. M., Lister, C. A., Snith, S. A., and Thurlby, P. L., [[ω-(Heterocyclylamino)alkoxy.]benzyl]-2,4-thiazolidinediones as potent antihyperglycemic agents.J. Med. Chem., 37, 3977–3985 (1994).
Green, L. C., Wagner, D. A., Glogowski, J., Skipper, P. L., Wishnok, J. S., and Tannenbaum, S. R., Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids.Anal. Biochem., 126, 131–138 (1982).
Heneka, M. T., Feinstein, D. L., Galea, E., Gleichmann, M., Wullner, U., and Klockgether, T., Peroxisome proliferator-activated receptor gamma agonists protect cerebellar granule cells from cytokine-induced apoptotic cell death by inhibition of inducible nitric oxide synthase.Journal of Neuroimmunology, 100, 156–168 (1999).
Henke, B. R., Blanchard, S. G., Brackeen, M. F., Brown, K. K., Cobb, J. E., Collins, J. L. Jr., Harrington, W. W., Hashim, M. A., Hull-Ryde, E. A., Kaldor, I., Kliewer, S. A., Lake, D. H., Leesnitzer, L. M., Lehmann, J. M., Lenhard, J. M., Orband-Miller, L. A., Miller, J. F., Mook, R. A. Jr., Noble, S. A., Oliver, W. Jr., Parks, D. J., Plunket, K. D., Szewczyk, J. R., and Willson, T. M., N-(2-benzoylphenyl)-L-tyrosine PPARgamma agonists. 1. Discovery of a novel series of potent antihyperglycemic and antihyperlipidemic agents.J. Med. Chem., 41, 5020–5036 (1998).
Jeon, R. and Park, S., Synthesis and biological activity of benzoxazole containing thiazolidinedione derivatives.Arch. Pharm. Res., 27, 1099–1105 (2004).
Lehmann, J. M., Moorre, L. B., Smith-Oliver, T. A., Wilkinson, W. O., Wilson, T. M., and Kliewer, S. A., An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor γ (PPARγ).J. Biol. Chem., 270, 12953–12956 (1995).
Lohray, B. B., Bhushan, V., Rao, B. P., Madhavan, G. R., Murali, N., Rao, K. N., Reddy, A. K., Rajesh, B. M., Reddy, P. G., Chakrabarti, R., Vikramadithyan, R. K., Rajagopalan, R., Mamidi, R. N., Jajoo, H. K., and Subramaniam, S., Novel euglycemic and hypolipidemic agents. 1.J. Med. Chem., 41, 1619–1630 (1998).
Morita, H., Mori, H., and Kobayashi, Y., Process for the production of a thiazolidinedione derivative, GB 2324089A (1998).
Oguchi, M., Wada, K., Honma, H., Tanaka, A., Kaneko, T., Sakakibara, S., Ohsumi, J., Serizawa, N., Fujiwara, T., Horikoshi, H., and Fujita, T., Molecular design, synthesis, and hypoglycemic activity of a series of thiazolidine-2,4-diones.J. Med. Chem., 43, 3052–3066 (2000).
Reaven, G. M., Chang, H., and Hoffman, B. B., Additive hypoglycemic effects of drugs that modify free-fatty acid metabolism by different mechanisms in rats with streptozocin-induced diabetes.Diabetes, 37, 28–32 (1988).
Sohda, T., Momose, Y., Meguro, K., Kawamatsu, Y., Sugiyama, Y., and Ikeda, H., Studies on antidiabetic agents. Synthesis and hypoglycemic activity of 5-[4-(pyridylalkoxy)benzyl]-2,4-thiazolidinediones.Arzneim.-Forsch., 40, 37–42 (1990).
Yoshioka, T., Fujita, T., Kanai, T., Aizawa, Y., Kurumada, T., Hasegawa, K., and Horikoshi, H., Studies on hindered phenols and analogues. 1. Hypolipidemic and hypoglycemic agents with ability to inhibit lipid peroxidation.J. Med. Chem., 32, 421–428 (1989).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeon, R., Kim, YJ., Cheon, Y. et al. Synthesis and biological activity of [[(heterocycloamino)alkoxy] benzyl]-2,4-thiazolidinediones as PPARγ agonists. Arch Pharm Res 29, 394–399 (2006). https://doi.org/10.1007/BF02968589
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02968589