Abstract
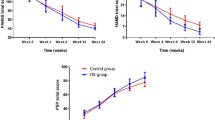
Results of a subanalysis of data from the multinational risperidone trial (RIS-INT-2) are reported. Patients with chronic schizophrenia were treated with risperidone at 1 mg/day (n = 25), 4 mg/day (n = 27), 8 mg/day (n = 29), 12 mg/day (n = 31), or 16 mg/day (n = 29), or 10 mg/day of haloperidol for 8 weeks. According to the Positive and Negative Syndrome Scale (PANSS) total and subscale scores, improvements were noted in each treatment group from baseline to treatment endpoint. On each scale the magnitude of improvement was greater in the risperidone patients than in the haloperidol patients. The onset of action of risperidone was faster than haloperidol. By treatment week 2, over half of the patients receiving ≥ 4 mg/day of risperidone were clinically improved (≥ 20% reduction in total PANSS scores). This rate of improvement was not seen until week 6 in the haloperidol patients. Severity of extrapyramidal symptoms (scores on the Extrapyramidal Symptom Scale) was significantly lower in patients receiving 1 or 4 mg/day of risperidone than in patients receiving higher risperidone doses and in haloperidol patients. The optimal dose of risperidone, in terms of both efficacy and safety, was 4 mg/day. These results confirm the findings of the controlled trials of risperidone conducted in North America and the multinational trial.
Similar content being viewed by others
References
American Psychiatric Association (1987) Diagnostic and statistical manual of mental disorders (DSM-III-R), 3rd edn, revised. American Psychiatric Press, Washington DC
Bersani G, Bressa GM, Meco G, Marini S, Pozzi F (1990) Combined serotonin 5-HT2 and dopamine-D2 antagonism in schizophrenia; clinical, extrapyramidal and neuroendocrine response in a preliminary study with risperidone (R 64 766) Hum Psychopharmacol 5: 225–231
Castelao JF, Ferreira L, Gelders YG, Heylen SLE (1989) The efficacy of the D2 and 5-HT2 antagonist risperidone (R 64 766) in the treatment of chronic psychosis: an open dose-finding study. Schizophr Bull 2: 411–415
Chouinard G, Ross-Chouinard A, Annable L, Jones BD (1980) The Extrapyramidal Symptom Rating Scale. Can J Neurol Sci 7: 233
Chouinard G, Jones B, Remington G, Bloom D, Addington D, MacEwan GW, Labelle A, Beauclair L, Arnott W (1993) A Canadian multicenter placebo-controlled study of fixed doses of risperidone and haloperidol in the treatment of chronic schizophrenic patients. J Clin Psychopharmacol 13: 25–40
Claus A, Bollen J, Cuyper H de, Eneman M, Malfroid M, Peuskens J, Heylen S (1992) Risperidone versus haloperidol in the treatment of chronic schizophrenic patients: a multicenter double-blind comparative study. Acta Psychiatr Scand 85: 275–305
Gelders YG (1989) Thymosthenic agents, a novel approach in the treatment of schizophrenia. Br J Psychiatry 155 (Suppl 5): 33–36
Gelders YG, Heylen SLE, Vanden Bussche G, Reyntjens AJM, Janssen PAJ (1990) Pilot clinical investigation of risperidone in the treatment of psychotic patients. Pharmacopsychiatry 23: 206–211
Guy W (1976) ECDEU Assessment manual for psychopharmacology (revised). Published by DHEW, no. (ADM) 76-338, Rockville, Maryland: National Institutes of Mental Health
Janssen PAJ, Niemegeers CJE, Awouters F, Schellekens KHL, Megens AA, Meert TF (1988) Pharmacology of risperidone (R64766), a new antipsychotic with serotonin-S2 and dopamine-D2 antagonistic properties. J Pharmacol Exp Ther 244: 685–693
Kay SR, Fisbein A, Opler LA (1987) The Positive and Negative Syndrome Scale (PANSS) for Schizophrenia. Schizophr Bull 13: 261–276
Leysen JE, Gommeren W, Eens A, De Chaffoy de Courcelles D, Stoof JC, Janssen PAJ (1988) The biochemical profile of risperidone, a new antipsychotic. J Pharmacol Exp Ther 247: 661–670
Linghaerde O, Ahlfors UG, Bech P, Dencker SJ, Eigen K (1987) The UKU side effect rating scale. Acta Psychiatr Scand 76 (Suppl 334): 81–94
Marder SR, Meibach RD (1995) Risperidone in the treatment of schizophrenia. Am J Psychiatry 151: 825–835
Marder SR, Davis JM, Chouinard G (1996) The clinical actions of risperidone. Poster presented at the annual meeting of the American College of Neuropsychopharmacology, 9–13 December, San Juan, Puerto Rico
Meco G, Bedini L, Bonifati V, Sonsini U (1989) Risperidone in the treatment of chronic schizophrenia with tardive dyskinesia: a single-blind crossover study versus placebo. Curr Ther Res 46: 876–883
Megens AA, Awouters FHL, Niemegeers CJE (1988) Differential effects of the new antipsychotic risperidone on large and small motor movements in rats: a comparison with haloperidol. Psychopharmacology 95: 493–496
Mesotten F, Suy E, Pietquin M, Burton P, Heylen S, Gelders Y (1989) Therapeutic effect and safety of increasing doses of risperidone (R 64 766) in psychotic patients. Psychopharmacology 99: 445–449
Möller HJ, Pelzer E, Kissling W, Riehl T, Wernicke T (1991) Efficacy and tolerability of a new antipsychotic compound (risperidone): results of a pilot study. Pharmacopsychiatry 24: 185–189
Overall JF, Gorham DR (1962) The Brief Psychiatric Rating Scale. Psychol Rep 10: 799–812
Peuskens J (1995) On behalf of The Risperidone Study Group. Risperidone in the treatment of chronic schizophrenic patients: a multi-national, multi-centre, double-blind, parallel-group study versus haloperidol. Br J Psychiatry 166: 712–726
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Möller, H.J., Bäuml, J., Ferrero, F. et al. Risperidone in the treatment of schizophrenia: results of a study of patients from Germany, Austria, and Switzerland. Eur Arch Psychiatry Clin Neurosci 247, 291–296 (1997). https://doi.org/10.1007/BF02922257
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02922257