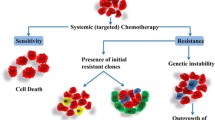
Conclusion
Considerable progress has been made toward understanding some of the molecular mechanisms underlying MDR in cancer cells in vitro, and sensitive techniques such as immunocytochemistry and RT-PCR indicate that these mechanisms may also play a role in resistance in human cancers. It does seem, however, that there are many different patterns and mechanisms of MDR, not all of which are currently well understood. Identification of chemicals which, at non-toxic doses, circumvent MDR, and of new drugs to which MDR cells are not cross resistant, remains a priority in this area of research. Also, it is important to remember that, although inherent resistance at the molecular level is possibly the most serious barrier to successful chemotherapy, other issues including tumor cell kinetics, drug metabolism, drug penetration within the tumor, and side effects on normal tissues are also critical factors in determining how an individual may respond to chemotherapy.
Similar content being viewed by others
References
Ali-Osman, F.; stein, D.; Renwick, A. Glutathione content and GST expression in BCNU-resistant human malignant astrocytoma cell lines. Cancer Res. 50:6976–6980; 1990.
Alon, N.; Busche, R.; Tummler, B., et al. Membrane lipids of MDR cells: chemical composition and physical state. In: Roninson, I., ed. Molecular biology of MDR in tumour cells. New York: Plenum Press; 1991:263–278.
Ariyoshi, K.; Hamada, H.; Naito, M., et al. Mouse-human chimeric antibody MH171 against the multidrug transporter P-glycoprotein. Jpn. J. Cancer Res. 83:515–521; 1992.
Baas, F.; Jongsma, A. P.; Broxterman, H. J., et al. Non-P-glycoprotein mediated mechanism for MDR precedes P-glycoprotein expression duringin vitro selection for doxorubicin resistance in a human lung cancer cell line. Cancer Res. 50:5392–5398; 1990.
Barrand, M. A.; Twentyman, P. R. Differential recognition ofmdr-1a andmdr-1b gene products in MDR mouse tumour cell lines by different monoclonal antibodies. Br. J. Cancer 65:239–245; 1992.
Batist, G.; Tulpule, A.; Sinha, B., et al. Overexpression of a novel anionic glutathione transferase in MDR human breast cancer cells. J. Biol. Chem. 261:15544–15549; 1986.
Beck, W. T.; Unknotting the complexities of multidrug resistance: the involvement of DNA topoisomerses in drug action and resistance. JNCI 81:1683–1685; 1989.
Becker, K. F.; Allmeier, H.; Hollt, V. New mechanisms of hormone secretion; MDR-like gene products as extrusion pumps for hormones. Horm. Metab. Res. 24:210–213; 1992.
Bell, D. R.; Trent, J. M.; Willard, H. F., et al. Chromosomal location of human P-glycoprotein gene sequences. Cancer Genet. Cytogenet. 25:141–148; 1987.
Bellamy, W. T.; Dalton, W. S.; Dorr, R. T. The clinical relevance of multidrug resistance. Cancer Invest. 8:547–562; 1990.
Bodley, A. L.; Liu, L. F. Topoisomerases as novel targets for cancer chemotherapy. Biotechnology 6:1315–1319; 1988.
Boscoboinik, D.; Gupta, R. S.; Epand, R. H. Investigation of the relationship between altered intracellular pH and MDR in mammalian cells. Br. J. Cancer 61:568–572; 1990.
Bradley, G.; Juranka, P. F.; Ling, V. Mechanisms of multidrug resistance. Biochim. Biophys. Acta 948:87–128; 1988.
Bremer, S.; Hoof, T.; Wilke, M., et al. Quantitative expression patterns of MDR P-glycoprotein (MDR1) and differentially spliced cystic fibrosis transmembrane conductance regulator mRNA transcripts in human epithelia. Eur. J. Biochem. 206:137–149; 1992.
Bugg, B. Y.; Danks, M. K.; Beck, W. T., et al. Expression of a mutant DNA topoisomerase II in CCRF-CEM human leukemic cells selected for resistance to teniposide. Proc. Natl. Acad. Sci. USA 88:7654–7658; 1991.
Burt, R. K.; Thorgeirsson, S. Co-induction ofmdr-1 multidrug resistance and cytochrome P-450 genes in rat liver by xenobiotics. JNCI 80:1383–1386; 1988.
Campbell, E.; Takashahi, Y.; Abramovitz, M., et al. A distinct human testis and brainμ-class glutathione S-transferase. J. Biol. Chem. 265:9188–9193; 1990.
Campbell, J.; Corrigall, A.; Guy, A., et al. Immunohistologic localisation of alpha, mu and pi class glutathione S-transferases in human tissues. Cancer 67:1608–1613; 1991.
Chao, C. C.; Huang, Y. T.; Ma, C. M., et al. Overexpression of GST and elevation of thiol pools in a MDR human colon cancer cell line. Mol. Pharmacol. 41:69–75; 1992.
Chen, C.; Clark, D.; Ueda, K., et al. Genomic organisation of the human MDR (MDR1) gene and origin of P-glycoproteins. J. Biol. Chem. 265:506–514; 1990.
Chin, J. E.; Soffir, R.; Noonan, K. E., et al. Structure and expression of the human MDR (P-glycoprotein) gene family. Mol. Cell Biol. 9:3808–3820; 1989.
Chin, K.; Tanaka, S.; Darlington, G., et al. Heat shock and arsenite increase expression of the MDR (MDR1) gene in human renal carcinoma cells. J. Biol. Chem. 265:221–226; 1990.
Chin, K.; Ueda, K.; Pastan, I., et al. Modulation of activity of the promoter of the human MDR1 gene by ras and p53. Science 255:459–462; 1992.
Choi, K.; Chen, C. J.; Kriegler, M., et al. An altered pattern of cross-resistance in MDR human cells results from spontaneous mutations in themdr-1 (P-glycoprotein) gene. Cell 53:519–529; 1988.
Clynes, M.; Redmond, A.; Heenan, M. Recent developments in research on multiple drug-resistance in cancer cells. Cancer J. 3:34–39; 1990.
Clynes, M.; Redmond, A.; Moran, E., et al. Multiple drug resistance in a human non-small cell lung carcinoma line DLKP-A. Cytotechnology. 10:75–89; 1992a.
Clynes, M.; Redmond, A.; Moran, E. Studies on MDR variants of human lung carcinoma lines. In Vitro Cell. Dev. Biol. 28:71A; 1992b.
Cohen, D.; Yang, C.; Horwitz, S. B. The products of themdr-1a andmdr-1b genes from MDR murine cells have similar degradation rates. Life Sci. 46:489–495; 1990.
Cole, S. P.; Downes, H. F.; Mirski, S. E., et al. Alterations in glutathione and glutathione-related enzymes in a MDR small cell lung cancer cell line. Mol. Pharmacol. 37:192–197; 1990.
Cole, S. P.; Chanda, E. G.; Dicke, F. P., et al. Non-P-glycoprotein-mediated MDR in a small cell lung cancer cell line: evidence for decreased susceptibility to drug-induced DNA damage and reduced levels of topoisomerase II. Cancer Res. 51:3345–3352; 1991.
Cole, S. P.; Pinkoski, M. J.; Bhardwaj, G., et al. Elevated expression of Annexin II (Lipocortin II, p. 36) in a multidrug resistant small cell lung cancer cell line. Br. J. Cancer 65:498–502; 1992a.
Cole, S. P.; Bharadwaj, G.; Gerlach, J. H., et al. Overexpression of a transporter gene in a multidrug-resistant human lung cancer cell line. Science 258:1650–1654; 1992b.
Coley, H. M.; Workman, P.; Twentyman, P. Retention of activity by selected anthracyclines in MDR human large cell carcinoma line without P-glycoprotein hyperexpression. Br. J. Cancer 63:351–357; 1991.
Collins, F. S. Cystic fibrosis: molecular biology and therapeutic implications. Science 256:774–779; 1992.
Cornwell, M. M. The human MDR gene: sequences upstream and downstream of the initiation site influence transcription. Cell Growth Differ. 1:607–615; 1990.
Croop, J. M.; Guild, B. C.; Gros, P., et al. Genetics of MDR: relationship of a cloned gene to the complete MDR Phenotype. Cancer Res. 47:5982–5988; 1987.
Croop, J. M.; Raymond, M.; Haber, D., et al. The 3 mouse multidrug resistance MDR genes are expressed in a tissue-specific manner in normal mouse tissues. Mol. Cell. Biol. 9:1346–1350; 1989.
Danks, M. K.; Schmidt, C. A.; Cirtain, M. C., et al. Altered catalytic activity of and DNA cleavage by DNA topoisomerase II from human leukemic cells selected for resistance to VM-26. Biochemistry 27:8861–8869; 1988.
Danks, M. K.; Schmidt, C. A.; Deneka, D. A., et al. Increased ATP requirement for activity of and complex formation by DNA topoisomerase II from human leukemic CCRF-CEM cells selected for resistance to teniposide. Cancer Commun. 1:101–109; 1989.
Debenham, P. G.; Kartner, N.; Siminovitch, L., et al. DNA-mediated transfer of MDR and plasma membrane glycoprotein expression. Mol. Cell. Biol. 2:881–889; 1982.
De Jong, S.; Zijlstra, J. G.; de Vries, E. G., et al. Reduced DNA topoisomerase II activity and drug-induced DNA cleavage activity in an adriamycin-resistant human small cell lung carcinoma cell line. Cancer Res. 50:304–309; 1990.
Devine, S. E.; Ling, V.; Melera, P. W. Amino acid substitutions in the 6th transmembrane domain of P-glycoprotein alter MDR. Proc. Natl. Acad. Sci. USA 89:4564–4568; 1992.
Dimanche-Boitrel, M.; Pelletier, H.; Genne, P., et al. Confluence-dependent resistance in human colon cancer cells: role of reduced drug accumulation and low intrinsic chemosensitivity of resting cells. Int. J. Cancer 50:677–682; 1992.
Doehmer, J.; Oesch, F. V79 Chinese hamster cells genetically engineered for stable expression of cytochromes P450. Methods Enzymol. 206:117–123; 1991.
Dupuis, M. L.; Ramoni, C.; Yassen, A., et al. The over-expression of P-glycoprotein in K-562 and Daudi cells is associated with a high susceptibility to NK and LAK cells. J. Biol. Reg. Homeostat. Agents 5:137–141; 1991.
Dudler, R.; Hertig, C. Structure of an mdr-like gene from arabidopsis thaliana—evolutionary implications. J. Biol. Chem. 267:5882–5888; 1992.
Dusre, L.; Mimnaugh, E. G.; Myers, C. E., et al. Potentiation of doxorubicin cytotoxicity by BSO in MDR human breast tumour cells. Cancer Res. 49:511–515; 1989.
Eijdems, E. W.; Borst, P.; Jongsma, A. P., et al. Genetic transfer of non-P-glycoprotein-mediated MDR in somatic cell fusion—dissection of a compound MDR phenotype. Proc. Nat. Acad. Sci. USA 89:3498–3502; 1992.
Endicott, J. A.; Ling, V. The biochemistry of P-glycoprotein-mediated multidrug resistance. Ann. Rev. Biochem. 58:137–171; 1989.
Fairchild, C. R.; Moscow, J. A.; O’Brien, E. E., et al. Multidrug resistance in cells transfected with human genes encoding a variant P-glycoprotein and glutathione-S-transferase pi. Mol. Pharmacol. 37:801–809; 1990.
Fischer, D. S.; Knobf, M. T. The cancer chemotherapy handbook. Chicago: Year Book Medical Publishers; 1989.
Foglesong, P. D.; Reckord, C.; Swink, S. Doxorubicin inhibits human DNA topoisomerase I. Cancer Chemother. Pharmacol. 30:123–125; 1992.
Ford, J. M.; Hait, W. N. Pharmacology of drugs that alter MDR in cancer. Pharmacol. Rev. 42:155–199; 1990.
Ford, J. M.; Yang, J.; Hait, W. N. Effect of BSO on toxicity of verapamil and doxorubicin to MDR cells and to mice. Cancer Res. 51:67–72; 1991.
Friche, E.; Danks, M. K.; Schmidt, C. A., et al. Decreased DNA topoisomerase II in daunorubicin-resistant Ehrlich ascites tumor cells. Cancer Res. 51:4213–4218; 1991.
Fry, A. M.; Chresta, C. M.; Davies, S. M., et al. Relationship between topoisomerase II level and chemosensitivity in human tumor cell lines. Cancer Res. 51:6592–6595; 1991.
Galski, H.; Sullivan, M.; Willingham, M. C., et al. Expression of a human MDR cDNA (MDR-1) in the bone marrow of transgenic mice: resistance to daunomycin-induced leukopenia. Mol. Cell. Biol. 9:4357–4363; 1989.
Georges, E.; Bradley, G.; Gariepy, J., et al. Detection of P-glycoprotein by gene-specific monoclonal antibodies. Proc. Natl. Acad. Sci. USA 87:152–156; 1990.
Giaccia, A. J.; Lewis, A. D.; Denko, N. C., et al. The hypersensitivity of the CHO variant BL-10 to bleomycin killing is due to a lack of GST alpha activity. Cancer Res. 51:4463–4469; 1991.
Giaccone, G.; Gazdar, A. F.; Beck, H., et al. Multidrug sensitivity phenotype of human lung cancer cells associated with topoisomerase II expression. Cancer Res. 52:1666–1674; 1992.
Gros, P.; Croop, J.; Housman, D. Mammalian MDR gene: complete cDNA sequence indicates strong homology to bacterial transport proteins. Cell 47:371–380; 1986.
Haber, M.; Reed, C.; Kavallaris, M., et al. Resistance to drugs associated with the MDR phenotype following selection with high-concentration methotrexate. JNCI 81:1250–1254; 1989.
Harris, A. L.; Hochhauser, D. Mechanisms of multidrug resistance in cancer treatment. Acta Oncol. 31:205–213; 1992.
Herweijer, H.; Sonnenveld, P.; Baas, F., et al. Expression ofmdr-1 and mdr-3 MDR genes in human acute and chronic leukemias in association with stimulation of drug accumulation by cyclosporine. JNCI 82:1133–1140; 1990.
Herzog, C. E.; Trepel, J. B.; Mickely, L. A., et al. Various methods of analysis ofmdr-1/P-glycoprotein in human colon cancer cell lines. JNCI 84:711–716; 1992.
Higgins, C. F.; Gottesman, M. M. Is the multidrug transporter a flippase? Trends Biochem. Sci. 17:18–21; 1992.
Hill, B. T.; Whelan, R. D.; Hosking, L. K., et al. A lack of detectable modification of topoisomerase II activity in a series of human tumour cell lines expressing only low levels of etoposide resistance. Int. J. Cancer 47:899–902; 1991.
Hoban, P. R.; Robson, C. N.; Davies, S. M., et al. Reduced topoisomerase II and elevated alpha class glutathione-S-transferase expression in a MDR CHO cell line highly cross-resistant to mitomycin C. Biochem. Pharmacol. 43:685–693; 1992.
Holzmayer, T. A.; Hilsenbeck, S.; Von Hoff, D. D., et al. Clinical correlates ofmdr-1 (P-glycoprotein) gene expression in ovarian and small cell lung carcinomas. JNCI. In press; 1992.
Hosking, L. K.; Whelan, R. D.; Shellard, S. A., et al. An evaluation of the role of glutathione and its associated enzymes in the expression of differential sensitivities to antitumour agents shown by a range of human tumour cell lines. Biochem. Pharmacol. 40:1833–1842; 1990.
Howie, A. F.; Forrester, L. M.; Glancey, M. J., et al. Glutathione-S-transferase and glutathione peroxidase expression in normal and tumour human tissues. Carcinogenesis 11:451–458; 1990.
Hsu, S. I.; Lothstein, L.; Horwitz, S. B. Differential overexpression of threemdr gene family members in MDR J774.2 mouse cells. J. Biol. Chem. 264:12053–12062; 1989.
Hsu, S. I.; Cohen, D.; Kirschner, L. S., et al. Structural analysis of mousemdr-1a (P-glycoprotein) promoter reveals the basis for differential transcript heterogeneity in MDR J774.2 cells. Mol. Cell. Biol. 10:3596–3606; 1990.
Huet, S.; Schott, B.; Robert, J. P-glycoprotein overexpression cannot explain the complete doxorubicin-resistance phenotype in rat glioblastoma cell lines. Br. J. Cancer 65:538–544; 1992.
Ichikawa, M.; Yoshimura, A.; Furukawa, T., et al. Glycosylation of P-glycoprotein in a MDR KB cell line, and in the human tissues. Biochim. Biophys. Acta. 1073:309–315; 1991.
Ivy, S. P.; Tulpule, A.; Fairchild, C. R., et al. Altered regulation of P4501A1 expression in a MDR MCF-7 human breast cancer cell line. J. Biol. Chem. 263:19119–19125; 1988.
Kamimoto, Y.; Gatmaitan, Z.; Hsu, J., et al. The function of gp 170, the MDR gene product, in rat liver canalicular membrane vesicles. J. Biol. Chem. 264:11693–11698; 1989.
Kamiwatari, M.; Nagata, Y.; Kikuchi, H., et al. Correlation between reversing of MDR and inhibiting3H-azidopine photolabelling of P-glycoprotein by newly-synthesized dihydropyridine analogues in a human cancer cell line. Cancer Res. 49:3190–3195; 1989.
Kaufmann, S. DNA topoisomerases in chemotherapy. Cancer Cells 3:24–27; 1991.
Keller, R. P.; Altermatt, H. J.; Nooter, K., et al. SDZ PSC-833, a non-immunosuppressive cyclosporine—its potency in overcoming P-glycoprotein-mediated MDR of human leukemia. Int. J. Cancer 50:593–597; 1992.
Kioka, N.; Tsubota, J.; Kakehi, Y., et al. P-glycoprotein gene (MDR-1) cDNA from human adrenal: normal P-glycoprotein carries Gly-185 with an altered pattern of multidrug resistance. Biochem. Biophys. Res. Commun. 162:224–231; 1989.
Lau, D. H.; Lewis, A. D.; Ehsan, M. N., et al. Multifactorial mechanisms associated with broad cross-resistance of ovarian carcinoma cells selected by cyanomorpholino doxorubicin. Cancer Res. 51:5181–5187; 1991.
Lee, S. A.; Karaszkiewicz, Anderson, W. B. Elevated level of nuclear protein kinase C in MDR MCF-7 human breast carcinoma cells. Cancer Res. 52:3750–3759; 1992.
Lewis, A. D.; Lau, D. H.; Duran, G. E., et al. Role of cytochrome P-450 from the human CYP3A gene family in the potentiation of morpholino doxorubicin by human liver microsomes. Cancer Res. 52:4379–4384; 1992.
Lincke, C. R.; van der Bliek, A.; Schuurhuis, G. J., et al. Multidrug resistance phenotype of human BRO melanoma cells transfected with a wild-type humanmdr-1 complementary DNA. Cancer Res. 50:1779–1785; 1990.
Lincke, C. R.; Smit, J.; Van der Velde-Koerts, T., et al. Structure of the humanMDR-3 gene and physical mapping of the humanMDR locus. J. Biol. Chem. 266:5303–5310; 1991.
Liu, L. F. DNA topoisomerase poisons as antitumor drugs. Ann. Rev. Biochem. 58:351–375; 1989.
Long, B. H.; Wang, L.; Lorico, A., et al. Mechanisms of resistance to etoposide and teniposide in acquired resistant human colon and lung carcinoma cell lines. Cancer Res. 51:5275–5284; 1991.
Mannervik, B. The enzymes of glutathione metabolism: an overview. Biochem. Soc. Trans. 15:717–719; 1987.
Marie, J. P.; Brophy, N. A.; Ehsan, M. N., et al. Expression of MDR genemdr-1 in a subset of normal marrow cells. Br. J. Haematol. 81:145–152; 1992.
Marquardt, D.; Center, M. S. Involvement of Vacuolar H+-ATPase activity in multidrug resistance in HL60 cells. JNCI 83:1098–1102; 1991.
Marquardt, D.; Center, M. S. Drug transport mechanisms in HL60 cells isolated for resistance to adriamycin—evidence for nuclear drug accumulation and redistribution in resistant cells. Cancer Res. 52:3157–3163; 1992.
Martin, A.; Clynes, M. Acid phosphatase: endpoint for in vitro toxicity tests. In Vitro Cell. Dev. Biol. 27A:183–184; 1991.
McGrath, J. P.; Varshavsky, A. The yeast STE6 gene encodes a homologue of the mammalian MDR P-glycoprotein. Nature 340:400–404; 1989.
McLachlin, J. R.; Eglitis, M. A.; Ueda, K., et al. Expression of a human cDNA for the MDR gene in murine hematopoietic precursor cells with the use of retroviral gene transfer. JNCI 82:1260–1263; 1990.
Mechetner, E. B.; Roninson, I. B. Efficient inhibition of P-glycoprotein-mediated MDR with a monoclonal antibody. Proc. Natl. Acad. Sci. USA 89:5824–5828; 1992.
Meese, E. V.; Horwitz, S. B.; Trent, S. M. Evidence for linear extrachromosomal elements mediating gene amplification in the MDR J774.2 murine cell line. Cancer Genet. Cytogenet. 59:20–25; 1992.
Michieli, M.; Damiani, D.; Geromin, A., et al. Overexpression of MDR-associated P-170 glycoprotein in acute non-lymphocytic leukemia. Eur. J. Haematol. 48:87–92; 1992.
Miyazaki, M.; Kohno, K.; Saburi, Y., et al. Drug resistance to cis-diamminedichloro-platinum (II) in CHO cell lines transfected with GST pi gene. Biochem. Biophys. Res. Commun. 166:1358–1364; 1990.
Moran, E.; Redmond, A.; Clynes, M. Preliminary studies of a monoclonal antibody raised to the over-expressed plasma membrane associated glycoprotein of a multidrug resistant cell line. Biochem. Soc. Trans. 20:58S; 1991.
Nakagawa K.; Saijo, N.; Tsuchida, S., et al. Glutathione-S-transferase pi as a determinant of drug resistance in transfectant cell lines. J. Biol. Chem. 265:4296–4301; 1990.
Negri, C.; Chiesa, R.; Cerino, A., et al. Monoclonal antibodies to human DNA topoisomerase I and the two isoforms of DNA topoisomerase II: 170- and 180-kDa isozymes. Exp. Cell Res. 200:452–459; 1992.
Ng, W. F.; Sarangi, F.; Zastawny, R. L., et al. Identification of members of the P-glycoprotein multigene family. Mol. Cell. Biol. 9:1224–1232; 1989.
Niehans, G. A.; Jaszcz, W.; Brunetto, V., et al. Immunohistochemical identification of P-glycoprotein in previously untreated, diffuse large cell and immunoblastic lymphomas. Cancer Res. 52:3768–3775; 1992.
Nooter, K.; Herweijer, H. Multidrug resistance (mdr) genes in human cancer. Br. J. Cancer 63:663–669; 1991.
Nygren, P.; Larsson, R.; Gruber, A., et al. Doxorubicin selected MDR small cell lung cancer cell lines characterised by elevated cytoplasmic Ca2+ and resistance modulation by verapamil in the absence of P-glycoprotein overexpression. Br. J. Cancer 64:1011–1018; 1991.
Ogura, M.; Takatori, T.; Sugimoto, Y., et al. Identification and characterisation of three DNA-binding proteins on the promoter of the humanMDR1 gene in drug-sensitive and -resistant cells. Jpn. J. Cancer Res. 82:1151–1159; 1991.
Ohtsu, T.; Ishido, Y.; Tobinai, K., et al. A novel MDR in cultured leukemia and lymphoma cells detected by a monoclonal antibody to 85 kDa protein, MRK20. Jpn. J. Cancer Res. 80:1133–1140; 1989.
Padmanabhan, A.; Tsuruo, T.; Kane, S., et al. Magnetic affinity sorting of human MDR cells. JNCI 83:565–575; 1991.
Peters, W. H. M.; Roelofs, H. M. J. Biochemical characterisation of resistance to mitoxantrone and adriamycin in CaCo-2 human colon adenocarcinoma cells—a possible role for gluthathione-S-transferases. Cancer Res. 52:1886–1890; 1992.
Philip, P. A.; Joel, S.; Monkman, S., et al. A phase I study on the reversal of MDRin vivo—nifedipine plus etoposide. Br. J. Cancer 65:267–270; 1992.
Pieters, R.; Hongo, T.; Loonen, A., et al. Different types of non-P-glycoprotein mediated MDR in children with relapsed acute lymphoblastic leukaemia. Br. J. Cancer 65:691–697; 1992.
Pratt, W. B.; Ridden, R. W. The anticancer drugs. New York: Oxford University Press; 1979.
Raymond, M.; Gros, P. Cell-specific activity of cis-acting regulatory elements in the promoter of the mouse MDR genemdr-1. Mol. Cell. Biol. 10:6036–6040; 1990.
Raymond, M.; Rose, E.; Houseman, D. E., et al. Physical mapping, amplification and overexpression of the mousemdr gene family in MDR cells. Mol. Cell Biol. 10:1642–1651; 1990.
Redmond, A.; Law, E.; Gilvarry, U., et al. Establishment of two MDR variants of the human tumor cell line Hep-2. Cell Biol. Toxicol. 6:293–300; 1990.
Redmond, A.; Moran, E.; Clynes, M. Analysis of the mechanism of resistance of 7 novel MDR variants. Biochem. Soc. Trans. 20:575; 1991.
Reeve, J. G.; Rabbitts, P. H.; Twentyman, P. R. Non-P-glycoprotein-mediated MDR with reduced EGF receptor expression in a human large cell lung cancer cell line. Br. J. Cancer 61:851–855; 1990.
Richardson, M. E.; Siemann, D. W. Thiol manipulation as a means of overcoming drug resistance in a novel cyclophosphamide-induced resistant cell line. Int. J. Radiat. Oncol. Biol. Phys. 22:781–784; 1992.
Rittmann-Grauer, L. S.; Yong, M. A.; Sanders, V., et al. Reversal of vinca alkaloid resistance by anti-p-glycoprotein monoclonal antibody HUB-241 in a human tumour xenograft. Cancer Res. 52:1810–1816; 1992.
Rivoltini, L.; Colomba, M.; Supino, R., et al. Modulation of MDR by verapamil orMDR1 antisense oligonucleotides does not change the high susceptibility to lymphokine-activated killers in MDR activated carcinoma (Lovo) line. Int. J. Cancer 46:727–732; 1990.
Roninson, I. B., editor. Molecular and cellular biology of multidrug resistance in tumour cells. New York: Plenum Publishing Corporation; 1991.
Roninson, I. B. The role ofMDR1 (P-glycoprotein) gene in MDRin vitro andin vivo. Biochem. Pharmacol. 43:95–102; 1992a.
Roninson, I. B. From amplification to function: the case of MDR1 gene. Mutat. Res. 276:151–161; 1992b.
Sakata, K.; Kwok, T. T.; Murphy, B. J., et al. Hypoxia-induced drug resistance—comparison to P-glycoprotein-associated drug resistance. Br. J. Cancer 64:809–814; 1991.
Schneider, J.; Efferth, T.; Mattern, J., et al. Immunohistochemical detection of the MDR marker P-glycoprotein in uterine cervical carcinomas and normal cervical tissue. Am. J. Obstet. Gynecol. 166:825–829; 1992.
Schoenlein, P. V.; Shen, D. W.; Barrett, J. T., et al. Double minute chromosomes carrying the humanmdr-1 and -2 genes are generated from the dimerization of submicroscopic circular DNAs in colchicine-selected KB carcinoma cells. Mol. Biol. Cell. 3:507–520; 1992.
Schurr, E.; Raymond, M.; Bell, J. C., et al. Characterisation of the MDR protein expressed in cell clones stably transfected with the mousemdr-1 cDNA. Cancer Res. 49:2729–2734; 1989.
Sehested, M.; Friche, E.; Jensen, P. B. Relationship of VP-16 to the classical MDR phenotype. Cancer Res. 52:2874–2879; 1992.
Sharma, R. C.; Inoue, S.; Roitelman, J., et al. Peptide transport by the MDR pump. J. Biol. Chem. 267:5731–5734; 1992.
Shen, D. W.; Lu, Y. G.; Chin, K. V., et al. Human hepatocellular carcinoma cell lines exhibit MDR unrelated to MDR1 gene expression. J. Cell Sci. 98:317–322; 1991.
Singh, S. V.; Nair, S.; Ahmad, H., et al. Glutathione-S-transferases and glutathione peroxidases in doxorubicin-resistant murine leukemic p388 cells. Biochem. Pharmacol. 38:3505–3509; 1989.
Sonneveld, P.; Durie, B.; Lokhorst, H., et al. Modulation of MDR multiple myeloma by cyclosporin. Lancet 340:255–259; 1992.
Stahl, F.; Wettergren, Y.; Levan, G. Amplicon structure in MDR murine cells—a nonrearranged region of genomic DNA corresponding to large circular DNA. Mol. Cell Biol. 12:1179–1187; 1992.
Sugimoto, Y.; Tsuruo, T. DNA-mediated transfer and cloning of a human MDR gene of adriamycin-resistant myelogenous leukemia K562. Cancer Res. 47:2620–2625; 1987.
Sullivan, D. M.; Latham, M. D.; Ross, W. E. Proliferation-dependent topoisomerase II content as a determinant of antineoplastic drug action in human, mouse and CHO cells. Cancer Res. 47:3973–3979; 1987.
Tan, K. B.; Dorman, T. E.; Falls, K. M., et al. Topoisomerase IIα and topoisomerase IIβ genes: characterisation and mapping to human chromosomes 17 and 3, respectively. Cancer Res. 52:231–234; 1992.
Taylor, C. W.; Dalton, W. S.; Parrish, P. R., et al. Different mechanisms of decreased drug accumulation in doxorubicin and mitoxantrone resistant variants of the MCF7 human breast cancer cell line. Br. J. Cancer 63:923–929; 1991.
Teeter, L. D.; Eckersberg, T.; Tsai, Y., et al. Analysis of the Chinese hamster P-glycoprotein/MDR genepgp1 reveals that the AP-1 site is essential for full promoter activity. Cell Growth & Differ. 2:429–437; 1991.
Tidefelt, U.; Elmhorn-Rosenburg, A.; Paul, C., et al. Expression of GST pi as a predictor for treatment results at different stages of acute nonlymphoblastic leukemia. Cancer Res. 52:3281–3285; 1992.
Toffoli, G.; Viel, A.; Tumiotto, L., et al. Expression of themdr-1 gene in human colorectal carcinomas—relationship with MDR inferred from analysis of human colorectal carcinoma cell lines. Cancer Chemother. Pharmacol. 29:283–289; 1992a.
Toffoli, G.; Viel, A.; Tumiotto, L., et al. Expression of GST-π in human tumours. Eur. J. Cancer 28A:1441–1446; 1992b.
Townsend, A. J.; Tu, C. F. D.; Cowan, K. H. Expression of human mu or alpha class GSTs in stably transfected human breast MCF-7 cancer cells—effect on cellular sensitivity to cytotoxic agents. Mol. Pharmacol. 41:230–236; 1992.
Valverde, M. A.; Diaz, M.; Sepulveda, F. V., et al. Volume-regulated chloride channels associated with the human MDR P-glycoprotein. Nature 355:830–833; 1992.
Van der Valk, P.; Van Kalken, C. K.; Ketelaars, H., et al. Distribution of MDR-associated P-glycoprotein in normal neoplastic human tissues; analysis with 3 monoclonal antibodies reorganizing different epitopes of the P-glycoprotein molecule. Ann. Oncol. 1:56–64; 1990.
Verelle, P.; Meissonnier, F.; Fonck, Y., et al. Clinical relevance of immunohistochemical detection of MDR P-glycoprotein in breast carcinoma. JNCI 83:111–116; 1991.
Versantvoort, C. H.; Broxterman, H. J.; Pineda, H. M., et al. Energy-dependent processes involved in reduced drug accumulation in MDR human lung cancer cell lines without P-glycoprotein expression. Cancer Res. 52:17–23; 1992.
Volm, M.; Mattern, J.; Samsel, B. Overexpression of P-glycoprotein and GST pi in resistant non-small cell lung carcinomas of smokers. Br. J. Cancer 64:700–704; 1991.
Wang, Y.; Teicher, B. A.; Shea, T. C., et al. Cross-resistance and GST-pi levels among 4 human melanoma cell lines selected for alkylating agent resistance. Cancer Res. 49:6185–6191; 1989.
Warrington, R. C.l-Histidinol in experimental cancer chemotherapy: improving the selectivity and efficacy of anticancer drugs, eliminating metastatic disease and reversing the MDR phenotype. Biochem. Cell Biol. 70:365–375; 1992.
Waxman, D. J. Glutathione-S-transferases: role in alkylating agent resistance and possible target for modulation chemotherapy—a review. Cancer Res. 50:6449–6454; 1990.
Webb, C. D.; Latham, M. D.; Lock, R. B., et al. Attenuated topoisomerase II content directly correlates with a low level of drug resistance in a CHO cell line. Cancer Res. 51:6543–6549; 1991.
Wolf, C. R.; Lewis, A. D.; Carmichael, J., et al. The role of glutathione in determining the response of normal and tumour cells to anticancer drugs. Biochem. Soc. Trans. 15:728–730; 1987.
Yang, C.; DePinho, S.; Greenberger, L., et al. Progesterone interacts with P-glycoprotein in MDR cells and in the endometrium of gravid uterus. J. Biol. Chem. 264:782–788; 1989.
Yang, L. Y.; Trujillo, J. M.. Biological characterisation of MDR human colon carcinoma sublines induced/selected by two methods. Cancer Res. 50:3218–3225; 1990.
Yoshimura, A.; Kuwazuru, Y.; Sumizawa, T., et al. Biosynthesis, processing and half-life of P-glycoprotein in a human MDR KB cell. Biochim. Biophys. Acta 992:307–314; 1989.
Zou, C. P.; Van, T. T.; Kuo, M. T. Isolation and characterisation of putative intrinsic MDR CHO cells by FACS. Anticancer Res. 12:427–432; 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Clynes, M. Cellular models for multiple drug resistance in cancer. In Vitro Cell Dev Biol - Animal 29, 171–179 (1993). https://doi.org/10.1007/BF02634176
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02634176