Summary
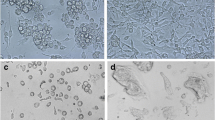
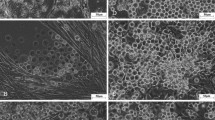
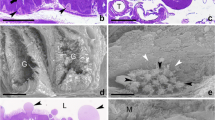
Midgut epithelial cells were isolated from fifth-instarPseudaletia unipuncta larvae by collagenase treatment of midgut tissue, and cultured in TNM-FH medium. Long-term continuous culture and maintenance of midgut cells were achieved withP. unipuncta armyworm intestinal cells. Several cells lines were obtained from theseP. unipuncta primary cultures, and they have been subcultured and maintained for over 24 mo. The three major midgut cell types were present in the cultures, including stem (regenerative), columnar, and goblet cells. In vitro morphogenesis and differentiation of columnar and goblet cells from stem cells were observed. There appeared to be a cycle of cell death of goblet and columnar cells followed by their replacement from stem cells every 7–8 wk. After approximately six passages, the cell density in T-flasks appeared to be somewhat constant, reaching 103–104 cells per milliliter of medium. The columnar cells are round to rectangular in shape and possess a brush border, while the goblet cells have a classic flask-like shape with a central cavity. Peritrophic membrane-like secretions were observed in all the culture flasks. Infection of these cells with multiply embedded nucleopolyhedrovirus was confirmed, and we conclude that these midgut cells can be used as an in vitro model system to study early events in baculovirus infection.
Similar content being viewed by others
References
Baines, D.; Brownwright, A.; Schwarrtz, J. L., Establishment of primary and continuous cultures of epithelial cells from larval lepidopteran midguts. J. Insect Physiol. 40:347–357; 1994.
Baines, D.; Schwarrtz, J. L.; Sohi, S. S.; Dedes, J.; Pang, A. Comparison of the response of midgut epithelial cells and cell lines from Lepidopteran larvae to CryIA toxins fromBacillus thuringiensis. J. Insect Physiol. 43:823–831; 1997.
Billingsly, P. F.; Lehane, M. J. Structure and ultrastructure of the insect midgut. In: Lehane, M. J.; Billingsly, P. F., ed. Biology of the insect midgut. London: Chapman and Hall; 1996:31–54.
Borchiellini, C.; Coulon, J.; Le, P. Y. The function of type IV collagen duringDrosophila muscle development. Mech. Dev. 58:179–191; 1996.
Braun, L.; Keddie, B. A. A new tissue technique for evaluating the effects ofBacillus thuringiensis toxins on insect midgut epithelium. J. Invertebr. Pathol. 69:92–104; 1997.
Engelhard, E. K.; Kam-Morgan, L. V.; Washburn, J. O.; Volkman, L. E. The insect tracheal system: a comduit for the systemic spread ofAutographa californica M nuclear polyhedrosis virus. Proc. Natl. Acad. Sci. USA 91:3224–3227; 1994.
Federici, B. A. Insecticidal bacterial proteins identify the midgut epithelium as a source of novel target sites for insect control. Arch. Insect Biochem. Physiol. 22:367–371; 1993.
Flint, N.; Cove, F. L.; Evans, G. S. Heparin stimulates the proliferation of intestinal epithelial cells in primary culture. J. Cell Sci. 107:401–411; 1994.
Girardi, A. J.; McMichael Jr., H.; Henle, W. The use of HeLa cells in suspension for the quantitative study of virus propagation. Virology 2:532–544; 1956.
Granados, R. R.; Lawler, K. A. In vivo pathway ofAutographa californica baculovirus invasion and infection. Virology 108:297–308; 1981.
Granados, R. R.; Li, G.; Derksen, C. G.; McKenna, K. A. A new insect cell line fromTrichoplusia ni (BTI-Tn-5B1-4) susceptible toTrichoplusia ni single enveloped nuclear polyhedrosis virus. J. Invertebr. Pathol. 64:260–266; 1994.
Granados, R. R.; Naughten, M. Replication ofAmsacta moorei entomopoxvirus andAutographa californica nuclear polyhedrosis virus in hemocyte cell lines fromEstigmene acrea. In: Kurstak, E.; Maramorosch, K., ed., Invertebrate tissue culture: applications in medicine, biology and agriculture. New York: Academic Press; 1976:379–384.
Granados, R. R.; Zhong, J.; Li, G.; Garcia, J.; Wang, P. Primary and established midgut cultures fromPseudaletia unipuncta andTrichoplusia ni larvae for baculovirus studies [abstract]. In Vitro Cell. Dev. Biol. 36(No. 3, Part II):15A; 2000.
Hink, W. F. Established insect cell line from the cabbage looper,Trichoplusia ni. Nature 220:466–467; 1970.
Kawai, Y.; Mitsuhashi, J. An insect cell line discrimination method by RAPD-PCR. In Vitro Cell. Dev. Biol. 33:512–515; 1997.
Kusche, G. M.; Garrison, K.; Mackrell, A. J.; Fessler, L. I.; Fessler, J. J. Laminin A chain: expression duringDrosophila development and genomic sequence. EMBO J. 11:4519–4527; 1992.
Lane, N. J.; Dallai, R.; Ashhurst, D. E. Structural macromolecules of the cell membranes and the extracellular matrices of the insect midgut. In: Lehane, M. J.; Billingsley, P. F., ed. Biology of the insect midgut. London: Chapman and Hall; 1996:115–150.
Loeb, M. J.; Hakim, R. S. Insect midgut epitheliumin vitro: an insect stem cell system. J. Insect Physiol. 42:1103–1111; 1996.
Loeb, M. J.; Hakim, R. S. Cultured midgut cells ofHeliothis virescens (Lepidoptera): fibronectin and integrin β 1 immunoreactivity during differentiationin vitro. Invertebr. Reprod. Dev. 35:95–102; 1999.
Loeb, M. J.; Hakim, R. S.; Martin, P.; Narang, N.; Goto, S.; Takeda, M. Apoptosis in cultured midgut cells fromHeliothis virescens larvae exposed to various conditions. Arch. Insect Biochem. Physiol. 45:12–23; 2000a.
Loeb, M. J.; Vaughn, J. L.; Clark, E. A. Primary cultures of midgut cells fromHeliothis virescens can be frozen and stored. In Vitro Cell. Dev. Biol. 36:7–10; 2000b.
Mitsuhashi, J.; Grace, T. D. C. The effects of insect hormones on the multiplication rates of cultured insect cellsin vitro. Appl. Entomol. Zool. 5:182–188; 1970.
Opas, M.; Dziak, E. Effects of substrata and method of tissue dissociation on adhesion cytoskeleton and growth of chick retinal pigmented epitheliumin vitro. In Vitro Cell. Dev. Biol. 24:885–892; 1988.
Sadrud-Din, S. Y.; Hakim, R. S.; Loeb, M. J. Proliferation and differentiation of midgut epithelial cells fromManduca sexta, in vitro. Invertebr. Reprod. Dev. 26:197–204; 1994.
Sadrud-Din, S. Y.; Loeb, M. J.; Hakim, R. S.In vitro differentiation of isolated stem cells from the midgut ofManduca sexta larvae. J. Exp. Biol. 199:319–325; 1996.
Vaughn, J. L.; Goodwin, R. H.; Tompkins, G. J.; McCawley, P. The establishment of two cell lines from the insectSpodoptera frugiperda (Lepidoptera: Noctuidae). In Vitro 13:213–217; 1977.
Wang, P.; Granados, R. R. An intestinal mucin is the target substrate for a baculovirus enhancin. Proc. Natl. Acad. Sci. USA 94:6977–6982; 1997.
Wang, P.; Granados, R. R.; Shuler, M. L. Studies on serum-free culture of insect cells for virus propagation and recombinant protein production. J. Invertebr. Pathol. 59:46–53; 1992.
Wang, P.; Hammer, D. A.; Granados, R. R. Interaction of enhancin, a viral encoded protein, from the granulosis virus ofTrichoplusia ni with the midgut epithelium and peritrophic membrane of four lepidopteran insects. J. Gen. Virol. 75:1961–1967; 1994.
Wang, S. M.; McCarthy, W. J. Cytolytic activity ofBacillus thuringiensis CrylC and CrylAc toxins toSpodoptera sp. midgut epithelial cellsin vitro. In Vitro Cell. Dev. Biol. 33:315–323; 1997.
Washburn, J. O.; Kirkpatrick, B. A.; Volkman, L. E. Insect protection against viruses. Nature 383:767; 1996.
Wyss, C.Chironomus tentans epithelial cell lines sensitive to ecdysteroids, juvenile hormone, insulin and heat shock. Exp. Cell Res. 139:309–319; 1982.
Yasothornsrikul, S.; Davis, W. J.; Cramer, G.; Kimbrell, D. A.; Dearolf, C. R. Viking. Identification and characterization of a second type IV collagen inDrosophila. Gene 198:17–25; 1997.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garcia, J.J., Li, G., Wang, P. et al. Primary and continuous midgut cell cultures fromPseudaletia unipuncta (lepidoptera: Noctuidae). In Vitro Cell.Dev.Biol.-Animal 37, 353–359 (2001). https://doi.org/10.1007/BF02577570
Issue Date:
DOI: https://doi.org/10.1007/BF02577570