Summary
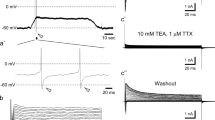
Apical membrane currents were recorded from the taste pore of single taste buds maintained in the tongue of the rat, using a novel approach. Under a dissection microscope, the 150-μm opening of a saline-filled glass pipette was positioned onto single fungiform papillae, while the mucosal surface outside the pipette was kept dry. Electrical responses of receptor cells to chemical stimuli, delivered from the pipette, were recorded through the pipette while the cells remained undamaged in their natural environment. We observed monophasic transient currents of 10-msec duration and 10–100 pA amplitude, apparently driven by action potentials arising spontaneously in the receptor cells. When perfusing the pipette with a solution of increased Na but unchanged Cl concentration, a stationary inward current (from pipette to taste cell) of 50–900 pA developed and the collective spike rate of the receptor cells increased. At a mucosal Na concentration of 250mm, the maximal collective spike rate of a bud was in the range of 6–10 sec−1. In a phasic/tonic response, the high initial rate was followed by an adaptive decrease to 0.5–2 sec−1. Buds of pure phasic response were also observed. Amiloride (30 μm) present in the pipette solution reversibly and completely blocked the increase in spike rate induced by mucosal Na. Amiloride also decreased reversibly the stationary current which depended on the presence of mucosal Na (inhibition constant near 1 μm). During washout of amiloride, spike amplitudes were first small, then increased, but always remained smaller than the amiloride-blockable stationary current of the bud. This is understandable since the stationary current of a bud arises from a multitude of taste cells, while each current spike is presumably generated by just one taste cell. We suggest that, in a Na-sensitive receptor cell, (i) the apical amiloride-blockable Na inward current serves as a generator current causing cell depolarization and firing of action potentials, and (ii) each current spike recorded from the taste pore arises mainly from a modulation of the apical Na inward current of this cell, because the action potential generated by the taste cell will transiently decrease or abolish the driving force for the apical Na inward current. The transients are indicators of receptor cell action potentials, which appear to be physiological responses of taste cellsin situ.
Similar content being viewed by others
References
Avenet, P., Lindemann, B. 1987. Action potentials in epithelial taste cells induced by mucosal Ca.J. Membrane Biol. 95:265–269
Avenet, P., Lindemann, B. 1988. Amiloride-blockable sodium currents in isolated taste receptor cells.J. Membrane Biol. 105:245–255
Avenet, P., Lindemann, B. 1989a. Chemoreception of salt taste. The blockage of stationary sodium currents by amiloride in isolated receptor cells and excised membrane patches.In: Chemical Senses: Molecular Aspects of Taste and Odor Reception. J.G. Brand, J.H. Teeter, R.H. Cagan, and M.R. Kare. editors. pp. 171–182. Marcel-Dekker, New York
Avenet, P., Lindemann, B. 1989b. Topical Review: Perspectives of taste reception.J. Membrane Biol. 112:1–8
Avenet, P., Lindemann, B. 1990. Fluctuation analysis of amiloride-blockable currents in membrane patches excised from salt-taste receptor cells.J. Basic Clin. Physiol. Pharmacol. 1 (Special issue):383–391
Avenet, P., Lindemann, B. 1991. A non-invasive method for recording action potentials of taste cells maintained in the tongue.Biophysical Society Annual Meeting, San Francisco (Abstr.)
Bartoshuk, L.M. 1974. NaCl thresholds in man: Thresholds for water taste or NaCl taste.J. Comp. Physiol. Psychol. 87:310–325
Behe, P., DeSimone, J.A., Avenet, P., Lindemann, B. 1989. Patch clamp recording from taste buds of maintained epithelial polarity: A novel approach. Xth International Symposium of Olfaction and Taste (ISOT) Oslo (Abstr.)
Behe, P., DeSimone, J.A., Avenet, P., Lindemann, B. 1990. Membrane currents in taste cells of the rat fungiform papilla: Evidence for two types of Ca currents and inhibition of K currents by saccharin.J. Gen. Physiol. 96:1061–1084
Brand, J.G., Teeter, J.H., Silver, W.L. 1985. Inhibition by amiloride of chorda tympani responses evoked by monovalent salts.Brain Res. 334:207–214
Delay, R.J., Kinnamon, J.C., Roper, S.D. 1986. Ultrastructure of mouse vallate taste buds: II. Cell types and cell lineage.J. Comp. Neurol. 253:242–252
Farbman, A.I. 1965. Fine structure of the taste bud.J. Ultrastruct. Res. 12:328–350
Faurion, A. 1982. Etude de mechanismes de la chimioreception du gout sucre.These de Doctorat d'Etat, Paris VI
Faurion, A., Vayssettes-Courchay, C. 1990. Taste as a highly discriminative system: A hamster intrapapillar single unit study with 18 compounds.Brain Res. 512:317–332
Forda, S.R., Jessel, T.M., Kelly, J.S., Rand, R.P. 1982. Use of the patch electrode for sensitive high resolution recording.Brain Res. 249:371–378
Frings, B., Lindemann, B. 1990. Single unit recording from olfactory cilia.Biophys. J. 57:1091–1094
Frings, S., Lindemann, B. 1991. Current recording from sensory cilia of olfactory receptor cellsin situ. I: The neuronal response to cyclic nucleotides.J. Gen. Physiol. 97:1–16
Heck, G.L., Mierson, S., DeSimone, J.A. 1984. Salt taste transduction occurs through an amiloride-sensitive sodium transport pathway.Science 223:403–405
Heck, G.L., Persaud, K.C., DeSimone, J.A. 1989. Direct measurement of translingual epithelial NaCl and KCl currents during the chorda tympani taste response.Biophys. J. 55:843–857
Kashiwayanagi, M., Miyake, M., Kurihara, K. 1983. Voltage-dependent Ca2+ and Na+ channel in frog taste cells.Am. J. Physiol. 244:C82-C88
Kinnamon, S.C., Dionne, V.E., Beam, K.G. 1988. Apical localization of K channels in taste cells provides basis for sour taste transduction.Proc. Natl. Acad. Sci. USA 85:7023–7027
Lapoint, J.-Y., Szabo, G. 1987. A novel holder allowing internal perfusion of patch pipettes.Pfluegers Arch. 410:212–216
Lindemann, B., Van Driessche, W. 1977. Sodium-specific membrane channels of frog skin are pores: Current fluctuations reveal high turnover.Science 195:292–294
Murray, R.G. 1971. Ultrastructure of taste receptors.In: Handbook of Sensory Physiology, L.H. Beidler, editor. Vol. IV, Part 2, pp. 31–50. Springer Verlag, Berlin-Heidelberg-New York
Richter, H.-P., Avenet, P., Mestres, P., Lindemann, B. 1988. Gustatory receptors and neighbouring cells in the surface layer of the frog's taste disc:in situ relationships and response to cell isolation.Cell Tissue Res. 254:83–96
Roper, S.D. 1983. Regenerative impulses in taste cells.Science 220:1311–1312
Roper, S.D., McBride, D.W. 1989. Distribution of ion channels on taste cells and its relationship to chemosensory transduction.J. Membrane Biol. 109:29–39
Taglietti, V., Casella, C., Ferrari, E. 1969. Interactions between taste receptors in the frog tongue.Pfluegers Arch. 312:139–148
Tang, J.M., Wang, J., Quandt, F.N., Eisenberg, R.S. 1990. Perfusing pipettes.Pfluegers Arch. 416:347–350
Yoshii, K., Kiyomoto, Y., Kurihara, K. 1986. Taste receptor mechanism of salts in frog and rat.Comp. Biochem. Physiol. 85A:501–507
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Avenet, P., Lindemann, B. Noninvasive recording of receptor cell action potentials and sustained currents from single taste buds maintained in the tongue: The response to mucosal NaCl and amiloride. J. Membrain Biol. 124, 33–41 (1991). https://doi.org/10.1007/BF01871362
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01871362