Summary
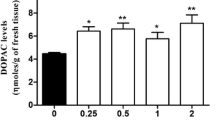
We measured the effect of ibogaine on the tritium efflux from isolated mouse striatum preloaded with [3H]dopamine ([3H]DA). Ibogaine increased the basal tritium outflow in a concentration-dependent manner, but it was without effect on electrical stimulation-induced tritium overflow. Separation of the released radioactivity after ibogaine administration showed that this drug increased the release of [3H]DA and [3H]-dihydroxyphenylacetic acid ([3H]DOPAC), but the efflux of O-methylated-deaminated metabolites was not changed. The dopamine (DA)-releasing effect of ibogaine was reduced by the DA uptake inhibitors cocaine and nomifensine. The tritium efflux evoked by ibogaine was not altered by omission of Ca2+ from the perfusion buffer or by inhibition of the voltage-sensitive Na+ channels with tetrodotoxin. Ibogaine maintained its effect on release from superfused striatum prepared from reserpine-pretreated mice. The ibogaine-induced tritium release measured from mouse striatum that was preloaded with [3H]DA was not affected by the D-2 DA receptor ligands (−)-quinpirole and (+/−)-sulpiride, indicating that the ibogaine-induced release is not subject to presynaptic autoreceptor regulation. Ibogaine failed to affect [3H]DA uptake and retention in mouse striatum. These data indicate that at the nerve terminal level ibogaine releases DA, and the primary source for the release is probably the cytoplasmic pool. The DA-releasing effect of ibogaine may have importance in mediation of its hallucinogenic action, as seen in a frequent practice in African cults.
Similar content being viewed by others
References
Adam-Vizi V (1992) External Ca2+-independent release of neurotransmitters. J Neurochem 58: 395–405
Broderick PA, Phelan FT, Paul Berger S (1991) Ibogaine alters cocaine-induced biogenic amine and psychostimulant dysfunction but not [3H]GBR-12935 binding to the dopamine transporter protein. Abst Comm Prob Drug Depend, NISA Res Monograph
Butcher SP, Fairbrother IS, Kelly JS, Arburnott GW (1988) Amphetamine-induced dopamine release in the rat striatum: an in vivo microdialysis study. J Neurochem 50: 346–355
Fairbrother IS, Arbuthnott GW, Kelly JS, Butcher SP (1990a) In vivo mechanisms underlying dopamine release from rat nigrostriatal terminals. I. Studies using veratrine and ouabain. J Neurochem 54: 1834–1843
Fairbrother IS, Arbuthnott GW, Kelly JS, Butcher SP (1990b) In vivo mechanisms underlying dopamine release from rat nigrostriatal terminals. II. Studies using potassium and tyramine. J Neurochem 54: 1844–1851
Glick SD, Rossman K, Steindorf S, Maisonneuve IM, Carlson JN (1991) Effects and after effects of ibogaine on morphine self-administration in rats. Eur J Pharmacol 195: 341–345
Harsing LG Jr, Sershen H, Lajtha A (1992a) Dopamine efflux from striatum after chronic nicotine: evidence for autoreceptor desensitization. J Neurochem 58: 48–54
Harsing LG Jr, Sershen H, Vizi ES, Lajtha A (1992b) N-type calcium channels are involved in the dopamine releasing effect of nicotine. Neurochem Res 17: 729–734
Kamal LA, Arbilla S, Langer SZ (1981) Presynaptic modulation of the release of dopamine from the rabbit caudate nucleus: differences between electrical stimulation, amphetamine and tyramine. J Pharmacol Exp Ther 216: 592–598
Lotsof H (1985) Rapid method for interrupting the narcotic addiction syndrome. Patent number 4, 499, 096
Lotsof H (1986) Rapid method for interrupting the cocaine and amphetamine abuse syndrome. Patent number 4, 587, 243
Lowry OH, Rosebrough N, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Maisonneuve IM, Keller RW Jr, Glick SD (1991) Interactions between ibogaine, a potential anti-additive agent, and morphine: an in vivo microdialysis study. Eur J Pharmacol 199: 35–42
Maisonneuve IM, Keller RW Jr, Glick SD (1992a) Interaction of ibogaine and d-amphetamine in rats: in vivo microdialysis and motor behavior in rats. Brain Res 579: 87–92
Maisonneuve IM, Rossman KL, Keller RW Jr, Glick SD (1992b) Acute and prolonged effects of ibogaine on brain dopamine metabolism and morphine-induced locomotor activity in rats. Brain Res 575: 69–73
Maisonneuve IM, Glick SD (1992) Interaction between ibogaine and cocaine in rats: in vivo microdialysis and motor behavior. Eur J Pharmacol 212: 263–266
Petride CE, Lombrazo L, Csernanszky JG (1990) A microassay for measuring synaptosomal3H-dopamine and3H-metabolite release. Brain Res Bull 25: 423–427
Raiteri M, Cerrito F, Cervoni A, Levi G (1979) Dopamine can be released by two mechanisms differentially affected by the dopamine transport inhibitor nomifensine. J Pharmacol Exp Ther 208: 195–202
Sershen H, Hashim A, Harsing L, Lajtha A (1992a) Ibogaine antagonizes cocaine-induced locomotor stimulation in mice. Life Sci 50: 1079–1086
Sershen H, Harsing LG Jr, Hashim A, Lajtha A (1992b) Ibogaine reduces amphetamine-induced locomotor stimulation in C57BL/6By mice, but stimulates locomotor activity in rats. Life Sci 51: 1003–1011
Sharpe L, Jaffe J (1992) Ibogaine fails to reduce naloxone-precipitated withdrawal in the morphine dependent rat. NIDA Research Monograph Series 105, pp477
Starke K, Gothert M, Kilbing H (1989) Modulation of neurotransmitter release by presynaptic autoreceptors. Physiol Rev 69: 864–989
Westerink BHC, Damsma G, Rollema H, De Vries JB, Horn AS (1987) Scope and limitations of in vivo brain dialysis: a comparison of its application to various neurotransmitter systems. Life Sci 41: 1736–1776
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Harsing, L.G., Sershen, H. & Lajtha, A. Evidence that ibogaine releases dopamine from the cytoplasmic pool in isolated mouse striatum. J. Neural Transmission 96, 215–225 (1994). https://doi.org/10.1007/BF01294788
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01294788