Abstract
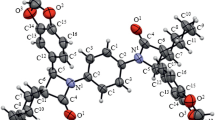
Stereospecific anti-addition of diethyl N,N-dibromophosphoramidate (DBPA) to (E)- and (Z)-2-butenes was definitely corroborated by X-ray structural determinations ofthreo- anderythro-2-amino-3-bromobutane hydrochlorides prepared from the corresponding adducts without changing the configuration on either chiral centers. The stereochemistry of azidation ofthreo- anderythro-2-amino-3-bromobutane hydrochlorides leading todl- andmeso-2,3-diaminobutane ditosylates respectively (after reduction of an azide function) was deduced from an X-ray crystallographic analysis of these diamine salts. The X-ray crystallographic analysis was carried out for three compounds: two reaction products and substrate (Scheme 2).
Similar content being viewed by others
References
Cahn, R. S., Ingold, C. K., Prelog, V. (1956)Experientia 12, 81. Cambridge Structural Database (1991).
Frenz, B. A.et al. (1984)SDP Structure determination package. (Enraf-Nonius, Delft, Holland).
Hanack, M. (1965)Conformation theory. (Academic Press, New York) p. 333.
International Tables for X-ray Crystallography (1974) (Kynoch Press, Birmingham, England).
Osowska-Pacewicka, K., and Zwierzak, A. (1985)Tetrahedron 41, 4717.
Osowska-Pacewicka, K., and Zwierzak, A. (1990)Synthesis 505.
Walker, N., and Stuart, D. (1983)Acta Crystallogr. A 39, 159.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wieczorek, M.W., Bujacz, G.D., Osowska-Pacewicka, K. et al. Stereochemistry of diamination of (E)- and (Z)-2-butenes: Crystal and molecular structures of meso- and dl-2,3-diaminobutane ditosylates. Journal of Crystallographic and Spectroscopic Research 23, 381–388 (1993). https://doi.org/10.1007/BF01159139
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01159139