Summary
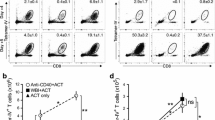
Survival of Fischer rats bearing 9L gliosarcoma in the brain was measured to determine the efficacy of 1) systemically administered chemotherapy with local adoptive immunotherapy (chemo-adoptive immunotherapy) or 2) systemically administered chemo-immunotherapy. Winn assays, where tumor instillation coincided with the start of treatment, and one-week established tumor assays were conducted. Survival of chemo-adoptive immunotherapy treated groups given intraperitoneal cyclophosphamide and intracranial lymphokine activated killer cells and recombinant Interleukin-2 was significantly extended when compared to sham treated control groups, to groups given chemotherapy with intraperitoneal cyclophosphamide, and to groups treated by local adoptive immunotherapy with intracranial lymphokine activated killer cells and Interleukin-2. The killer cells were generated from spleens of donor rats that either had or had not been given cyclophosphamide 24 h earlier. Long-term survivors (9/39), sacrificed at day 70, were obtained only in the chemo-adoptive immunotherapy treated groups; 7/39 had no histologic evidence of tumor and had focal sterile abscesses at the site of killer cell instillation. Average group weight plotted over time showed that there was acceptable toxicity with chemo-adoptive immunotherapy; the toxicity was identical to that obtained with systemic cyclophosphamide treatment. In contrast, survival of chemo-immunotherapy treated groups given systemic cyclophosphamide and Interleukin-2 was not significantly extended from groups which were sham treated or treated only with systemic Interleukin-2. Rapid decline of average group weight plotted over time and early deaths following chemo-immunotherapy treatment indicated that the regimen was toxic. The effect of cyclophosphamide administration on the splenocytes of donor rats and the LAK cells generated from them was determined byin vitro studies analyzing cell number, viability, phenotypic expression and cytotoxicity against 9L tumor. In the treatment of this intracranial neoplasm, the beneficial effects of cyclophosphamide were determined to occurin situ in the tumor-bearing host. No benefit resulted from cyclophosphamide treatment of donor rats that supplied splenocytes for LAK cell production.
Similar content being viewed by others
References
Kornblith PL, Walker MD, Cassady JR: Treatment of patients with supratentorial tumors-astrocytomas, oligodendrogliomas and optic gliomas. In: Kornblith PL, Walter MD, Cassady JR (eds) Neurologic Oncology. J.B. Lippincott Company, Philadelphia, 1987, pp 117–154
Robb RJ, Kutney RM, Chowdry V: Purification and partial sequence analysis of human T cell growth factor. Proc Natl Acad Sci (USA) 80: 5590–5595, 1983
Grimm EA, Mazumder A, Zhang HZ, Rosenberg SA: The lymphokine activated killer cell phenomena: lysis of NK resistant fresh solid tumor cells by IL-2 activated autologous human peripheral blood lymphocytes. J Exp Med 155: 1823–1841, 1982
Rosenberg SA, Lotze MT, Muul LM, Chang AE, Avid FP, Leitman S, Linehan M, Robertson CN, Lee RE, Rubin JT, Seipp CA, Simpson C, White DE: A progress report on the treatment of 157 patients with advanced cancer using lymphokine-activated killer cells and interleukin-2 or high dose interleukin-2 alone. New Engl J Med 316: 889–897, 1987
Lillehei KO, Kruse CA, Mitchell DH, Johnson SD, McCleary EL: Adoptive immunotherapy of recurrent glioma using Interleukin-2 stimulated lymphocytes. Surgical Forum 40: 493–495, 1989
Lillehei KO, Mitchell DH, Johnson SD, McCleary EL, Kruse CA: Long-term follow-up of patients with recurrent malignant gliomas treated with adjuvant adoptive immunotherapy. Neurosurgery 28: 16–23, 1991
Jacobs SK, Wilson DJ, Kornblith PL, Grimm EA: Interleukin-2 or autologous lymphokine-activated killer cell treatment of malignant glioma: phase I trial. Cancer Res 46: 2101–2104, 1986
Barba D, Saris SC, Holder C, Rosenberg SA, Oldfield EH: Intratumoral LAK cell and interleukin-2 therapy of human gliomas. J Neurosurg 70: 175–182, 1989
Merchant RE, Grant AJ, Merchant LH, Young HF: Adoptive immunotherapy for recurrent glioblastoma multiforme using lymphokine activated killer cells and recombinant Interleukin-2. Cancer 62: 665–671, 1988
Ingram M, Buckwalter JG, Jacques DBS, Freshwater DB, Abts RM, Techy GB, Miyagi K, Shelden CH, Rand RW, English LW: Immunotherapy for malignant glioma: an interim report on survival. Neurological Res 12: 265–273, 1990
Kitahara T, Watanabe O, Yamaura A, Makino H, Watanabe T, Suzuki G, Okumura K: Establishment of Interleukin-2 dependent cytotoxic T lymphocytes cell line specific for autologous brain tumor and its intracranial administration for therapy of the tumor. J Neuro-Oncology 4: 329–336, 1987
Yoshida S, Tanaka R, Takai N, Ono K: Adoptive immunotherapy with LAK cells and Interleukin-2 in the treatment of recurrent malignant gliomas. Current Therapeut Res 47: 654–665, 1990
Kempf RA, Mitchell MS: Effects of chemotherapeutic agents on the immune response. Cancer Investigation 2: 459–466, 1984
North RJ: Cyclophosphamide-facilitated adoptive immunotherapy of an established tumor depends on elimination of tumor-induced suppressor T cells. J Exp Med 55: 1063–1074, 1982
Berd D, Mastrangelo MJ: Effect of low dose cyclophosphamide on the immune system of cancer patients. Reduction of T-suppressor function without depletion of the CD8+ subset. Cancer Res 47: 3317–3321, 1987
Berd D, Mastrangelo MJ: Effect of low dose cyclophosphamide on the immune system of cancer patients: depletion of CD4+, 2H4+ suppressor-inducer T-cells. Cancer Res 48: 1671–1675, 1988.
Isaacs JT: Relationship between tumor size and curability of prostatic cancer by combined chemo-hormonal therapy in rats. Cancer Res 49: 6290–6294, 1989
Mitchell MS: Combining chemotherapy with biological response modifiers in treatment of cancer. J Natl Cancer Inst 80: 1445–1450, 1988
Kedar E, Ben-Aziz R, Epstein E, Leshem B: Chemoimmunotherapy of murine tumors using interleukin-2 (IL-2) and cyclophosphamide. Cancer Immunol Immunother 29: 74–78, 1989
Papa MZ, Yang JC, Vetto JT, Shiloni E, Eisenthal A, Rosenberg SA: Combined effects of chemotherapy and interleukin-2 in the therapy of mice with advanced pulmonary tumors. Cancer Res 48: 122–129, 1988
Lee K, O'Donnell RW, Marquis D, Cockett AT: Eradication of palpable intradermal murine bladder tumors by systemic interleukin-2 and cyclophosphamide in C3H mice. J Biol Response Mod 7: 32–42, 1988
Ootsu K, Gotoh K, Houkan T: Therapeutic efficacy of human recombinant interleukin-2 (TGP-3) alone or in combination with cyclophosphamide and immunocompetent cells in allogeneic, semi-syngeneic, and syngeneic murine tumors. Cancer Immunol Immunother 30: 71–80, 1989
Silagi S, Schaefer AE: Successful immunotherapy of mouse melanoma and sarcoma with recombinant interleukin-2 and cyclophosphamide. J Biol Response Mod 5: 411–422, 1986
Mule JJ, Rosenberg SA: Combination cytokine therapy: experimental and clinical trials. In: DiVita VTJr, Hellman S, Rosenberg SA (eds) Biologic Therapy of Cancer. J.B. Lippincott Co., Philadelphia, 1991, pp 393–416
Kedar E, Ben-Aziz R, Shiloni E; Therapy of advanced solid tumors in mice using chemotherapy in combination with interleukin-2 with and without lymphokine-activated killer cells. Israel J Med Sci 24: 494–504, 1988
Rosenberg SA, Spiess P, Lafreniere R: A new approach to the adoptive immunotherapy of cancer with tumor infiltrating lymphocytes. Science 233: 1318–1321, 1986
Eggermont AMM, Sugarbaker PH: Efficacy of chemoimmunotherapy with cyclophosphamide, interleukin-2 and lymphokine activated killer cells in an intraperitoneal murine tumour model. Br J Cancer 58: 410–414, 1988
Cheever MA, Greenberg PD, Fefer A: Specific adoptive therapy of established leukemia with syngeneic lymphocytes sequentially immunizedin vivo andin vitro and non-specifically expanded by culture with interleukin-2. J Immunol 126: 1318–1322, 1981
Cheever MA, Greenberg PD, Fefer A, Gillis S: Augmentation of the anti-tumor therapeutic efficacy of long-term cultured T lymphocytes byin vivo administration of purified interleukin 2. J Exp Med 155: 968–980, 1982
Greenberg PD: Adoptive T cell therapy of tumors: mechanisms operative in the recognition and elimination of tumor cells. Adv Immunol 49: 281–355, 1991
Kruse CA, Lillehei KO, Mitchell DH, Kleinschmidt-De-Masters BK, Bellgrau D: Analysis of interleukin-2 and various effector cell populations in adoptive immunotherapy of 9L rat gliosarcoma: allogeneic cytotoxic T lymphocytes prevent tumor take. Proc Natl Acad Sci (USA) 87: 9577–9581, 1990
Tzeng J-J, Barth RF, Clendenon NR, Gordon WA: Adoptive immunotherapy of a rat glioma using lymphokine-activated killer cells and interleukin-2. Cancer Res 50: 4338–4343, 1990
Yamasaki T, Handa H, Yamashita J, Watanabe Y, Namba Y, Hanaoka M: Specific adoptive immunotherapy with tumor-specific cytotoxic T lymphocyte clone for murine malignant gliomas. Cancer Res 44: 1776–1783, 1984
Takai N, Tanaka R, Yoshida S, Hara N, Saito T:In vivo andin vitro effect of adoptive immunotherapy of experimental murine brain tumors using lymphokine-activated killer cells. Cancer Res 48: 2047–2052, 1988
Mulvin DW, Kruse CA, Mitchell DH, Marcell T, James GT, Johnston M: Lymphokine activated killer cells with Interleukin-2: Dose toxicity and localization in isolated perfused rat lungs. Mol Biother 2: 38–43, 1990
Bellgrau D, Lagarde A-C: Cytotoxic T cell precursors with low-level CD8 in the diabetes-prone biobreeding rat: implications for generation of an autoimmune T-cell repertoire. Proc Natl Acad Sci (USA) 87: 313–317, 1990
Kruse CA, Mitchell DH, Lillehei KO, Johnson SD, McCleary EL, Moore GE, Waldrop S, Mierau GW: Interleukin-2 activated lymphocytes from brain tumor patients. A comparison of two preparations generatedin vitro. Cancer 64: 1629–1637, 1989
Ballas ZK: Lymphokine-activated killer (LAK) cells I. Differential recovery of LAK, natural killer cells, and cytotoxic T lymphocytes after a sublethal dose of cyclophosphamide. J Immunol 137: 2380–2384, 1986
Kim R, Lafreniere R, Borkenhagen K, Bryant LD: Induction of cytotoxicity from fresh splenocytes afterin vivo administration of cyclophosphamide. Cancer Immunol Immunother 30: 283–288, 1989
Cotran RS, Pober JS, Gimbrone MAJr, Springer TA, Wiebke EA, Gaspari AA, Rosenberg SA, Lotze MT: Endothelial activation during interleukin 2 immunotherapy. A possible mechanism for the vascular leak syndrome. J Immunol 139: 1883–1888, 1987
Rosenstein M, Ettinghausen SE, Rosenberg SA: Extravasation of intravascular fluid mediated by the systemic administration of recombinant interleukin 2. J Immunol 137: 1735–1742, 1986
Dropcho EJ, Mahaley MS: Chemotherapy for malignant gliomas in adults. In: Thomas DGT (ed) Neuro-oncology. Johns Hopkins University Press, Baltimore, 1990, pp 222–241
Yung WKA, Levin VA: Chemotherapy: current and future role and expectations. In: Apuzzo MLJ (ed) Malignant Cerebral Glioma. AANS Publications, Park Ridge, IL, 1990, pp 207–216
Powrie F, Mason D: Subsets of rat CD4+ T cells defined by their differential expression of variants of the CD45 antigen: developmental relationships andin vitro andin vivo functions. Curr Top Microbiol Immunol 159: 79–96, 1990
Merchant RE, Fontana A, Nelson ATJr, Young HF: Mitogen-activated lymphocytes of normals and glioma patients produce factors with anti-glioblastoma activityin vitro. J Neuroimmunol 11: 1–14, 1986
Atkinson LL, Merchant RE, Ghatak NR, Young HF: Sterile abscesses in glioma patients treated by intraparenchymal injection of lymphokine-activated killer cells and recombinant interleukin-2. Neurosurgery 25: 805–810, 1989
Kolitz JE, Wong GY, Weite K, Merluzzi VJ, Engert A, Bialas T, Polivka A, Bradley EC, Konrad M, Gnecco C, Oettgen HF, Mertelsmann R: Phase I trial of recombinant interleukin-2 and cyclophosphamide: augmentation of cellular immunity and T-cell mitogenic response with longterm administration of rIL-2. J Biol Response Mod 7: 457–472, 1988
Croghan M, Hersh EM, Taylor C, Meyskens F, Dalke P, McDonald L, Gardern S, Rudolph A: Phase I–II study of low dose cytoxan and recombinant interleukin-2 (IL-2) for the treatment of disseminated carcinoma (Abstract). Proc Am Soc Clin Oncol 8: 180, 1989
Rubin JT, Elwood LJ, Rosenberg SA, Lotze MT: Immunohistochemical correlates of response to recombinant interleukin-2 based immunotherapy in humans. Cancer Res 49: 7086–7092, 1989
Mitchell MS, Kempf RA, Harel W, Shau H, Boswell WD, Lind S, Bradley EC: Effectiveness and tolerability of lowdose cyclophosphamide and low-dose intravenous interleukin-2 in disseminated melanoma. J Clin Oncol 6: 409–424, 1988
Rosenberg SA: Clinical immunotherapy studies in the surgery branch of the U.S. National Cancer Institute: brief review. Cancer Treatment Reviews 16: 115–121, 1989
Topalian SL, Solomon D, Avis FP, Chang AE, Freerksen DL, Linehan WM, Lotze MT, Robertson CN, Seipp C, Simon P, Rosenberg SA: Immunotherapy of patients with advanced cancer using tumor infiltrating lymphocytes and recombinant interleukin-2: a pilot study. J Clin Oncol 6: 839–853, 1988
Kohler PC, Hank JA, Minkoff DG, Sondel PM: Clinical adoptive chemoimmunotherapy with allogeneic alloactivated HLA-haploidentical lymphocytes: controlled induction of graft-versus-host-reactions. Cancer Immunol Immunother 26: 74–82, 1988
Naganuma H, Kimurat R, Sasaki A, Fukamachi A, Nukui H, Tasaka K: Complete remission of recurrent glioblastoma multiforme following local infusions of lymphokine activated killer cells. Case report. Acta Neurochir (Wien) 99: 157–160, 1989
Nitta T, Sato K, Yagita H, Okumura K, Ishii S: Preliminary trial of specific targeting therapy against malignant glioma. The Lancet 335: 368–370, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kruse, C.A., Mitchell, D.H., Kleinschmidt-DeMasters, B.K. et al. Systemic chemotherapy combined with local adoptive immunotherapy cures rats bearing 9L gliosarcoma. J Neuro-Oncol 15, 97–112 (1993). https://doi.org/10.1007/BF01053931
Issue Date:
DOI: https://doi.org/10.1007/BF01053931