Conclusions
-
1.
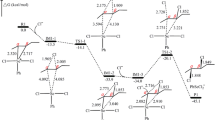
The catalytic dehydrochlorination of compounds of general formula CH3CCl2CH2CHClX (X = H, Cl, CH3) under the influence of Friedel-Crafts catalysts is accomplished via the chlorine of the dichloromethylene group toward both the methyl and the methylene groups.
-
2.
The alkaline dehydrochlorination of the same compounds proceeds with the involvement of the hydrogen of the methylene group and the chlorine of both chlorine-containing groups.
Similar content being viewed by others
Literature cited
R. Kh. Freidlina, T. T. Vasil'eva, R. G. Gasanov, P. V. Petrovskii, E. Ts. Chukovskaya, and N. A. Kuz'mina, Izv. Akad. Nauk SSSR, Ser. Khim., 1892 (1971).
R. Kh. Freidlina, T. T. Vasil'eva, V. I. Dostovalova, and E. Ts. Chukovskaya, Izv. Akad. Nauk SSSR, Ser. Khim., 2236 (1972).
V. N. Kost and R. Kh. Freidlina, Iav. Akad. Nauk SSSR, Otd. Khim. Nauk., 1253 (1961).
G. M. Shakhnazaryan, L. A. Saakyan, and M. T. Dangyan, Zh. Organ. Khim.,4, 1590 (1968).
A. N. Nesmeyanov, R. Kh. Freidlina, and V. I. Firstov, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 505 (1951).
R. Kh. Freidlina, V. I. Dostovalova, E. Ts. Chukovskaya, and T. T. Vasil'eva, Zh. Struktur. Khim.,14, 1011 (1973).
E. Ts. Chukovskaya, N. A. Kuz'mina, and R. Kh. Freidlina, Izv. Akad. Nauk SSSR, Ser. Khim., 2343 (1970); Dokl. Akad. Nauk SSSR,188, 1073 (1969).
T. Sato, M. Seno, and T. Asahara, Monthly J. Inst. Ind. Sci. Univ. Tokyo,24, No. 6, 230 (1972).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 3, pp. 622–627, March, 1974.
Rights and permissions
About this article
Cite this article
Vasil'eva, T.T., Sokolova, L.N., Dostovalova, V.I. et al. Dehydrochlorination of polychloroalkanes that contain the CH3CCl2CH2 CHCl group. Russ Chem Bull 23, 586–590 (1974). https://doi.org/10.1007/BF00921150
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00921150