Abstract
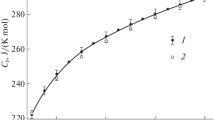
Acid strength of solid superacids was determined by temperature programmed desorption using pyridine. An approximately linear relationship exists between the acid strengths of solid acids determined by the Hammett method and the termination temperature of pyridine desorption. The acid strength of colored superacids versus temperature relationships practically fell on the linear line.
Similar content being viewed by others
References
K. Arata, Adv. Catal. 37 (1990) 165, and references therein.
K. Arata and M. Hino,Proc. 10th Int. Congr. on Catalysis, Budapest 1992, eds. L. Guczi, F. Solymosi and P. Tétényi (Akadémiai Kiadó, Budapest, 1993) p. 2613.
S. Lin and R. Hsu, J. Chem. Soc. Chem. Commun. (1992) 1469.
Z. Gao, J. Chen, W. Hua, and Y. Tang,Acid-Base Catalysis II, Int. Symp. in Honor of Kozo Tanabe, Sapporo, 2–4 December 1993, Prep. p. 26.
M. Niwa, M. Iwamoto and K. Segawa, Bull. Chem. Soc. Japan 59 (1986) 3735.
K. Arata and H. Sawamura, Bull. Chem. Soc. Japan 48 (1986) 3377.
H. Matsuhashi and K. Arata, Bull. Chem. Soc. Japan 64 (1991) 2605.
H. Matsuhashi, K. Kato and K. Arata,Acid-Base Catalysis II, Int. Symp. in Honor of Kozo Tanabe, Sapporo, 2–4 December 1993, Prep. p. 162.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Matsuhashi, H., Motoi, H. & Arata, K. Determination of acid strength of solid superacids by temperature programmed desorption using pyridine. Catal Lett 26, 325–328 (1994). https://doi.org/10.1007/BF00810605
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00810605