Abstract
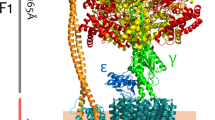
A molecular model for transmembrane segments 1 and 2 from the fungal proton pumping ATPase has been developed, and this structure is predicted to form a helical hairpin loop structure in the membrane. This region was selected because it is highly conformationally active and is believed to be an important site of action for clinically important therapeutics in related animal cell enzymes. The hairpin loop is predicted to form an asymmetric tightly packed structure that is stabilized by an N-cap between D140 and V142, by hydrogen bonding between residues in the turn region and the helices, and by π-π interactions between closely apposed aromatic residues. A short four-residue S-shaped turn is stabilized by hydrogen bonding but is predicted to be conformationally heterogeneous. The principal effect of mutations within the hairpin head region is to destabilize the local close packing of side groups which disrupts the pattern of hydrogen bonding in and around the turn region. Depending on the mutation, this causes either a localized or a more global distortion of the primary structure in the hairpin region. These altered structures may explain the effects of mutations in transmembrane segments 1 and 2 on ATP hydrolysis, sensitivity to vanadate, and electrogenic proton transport. The conformational sensitivity of the hairpin structure around the S-turn may also account for the effects of SCH28080 and possibly ouabain in blocking ATPase function in related animal cell enzymes. Finally, the model of transmembrane segments 1 and 2 serves as a template to position transmembrane segments 3 and 8. This model provides a new view of the H+-ATPase that promotes novel structure/function experimentation and could serve as the basis for a more detailed model of the membrane sector of this enzyme.
Similar content being viewed by others
References
Arystarkhova, E., Gasparian, M., Modyanov, N. N., and Sweadner, K. J. (1992).J. Biol. Chem. 19, 13694–13701
Bigelow, D. J., and Inesi, G. (1991).Biochemistry 30, 2113–2125.
Brandl, C. J., and Deber, C. M. (1986).Proc. Natl. Acad. Sci. USA 83, 917–921.
Canessa, C. M., Horisberger, J.-D., and Rossier, B. C. (1992).EMBO J. 11, 1681–1687.
Chou, K.-C., Maggiora, G. M., Nemethy, G., and Schegara, H. A. (1988).Proc. Natl. Acad. Sci. USA 85, 4295–4299.
DeGrado, W. F., Wasserman, Z. R., and Lear, J. D. (1989).Science 243, 622–628.
Eriksson, A. E., Baase, W. A., Zhang, X.-J., Heinz, D. W., Blaber, M., Blaldwin, E. P., and Mathews, B. W. (1992).Science 255, 178–183.
Feschenko, M. S., Zvaritch, E. I., Hofmann, F., Shakhparonov, M. I., Modyanov, N. N., Vorherr, T., and Carafoli, E. (1992).J. Biol. Chem. 267, 4097–4101.
Findlay, J. B. C., Eliopoulos, E. E., and Finbow, M. (1990).Biochem. Soc. Trans. 18, 838–840.
Forbush, B., III (1983).Curr. Top. Membr. Transp. 19, 167–201.
Glynn, I. M., and Karlish, S. J. D. (1990).Annu. Rev. Biochem. 59, 171–205.
Harris, S. L., Perlin, D. S., Seto-Young, D. and Haber, J. E. (1991).J. Biol. Chem. 266, 24439–24445.
Hennesey, J. P., and Scarborough, G. A. (1990).J. Biol. Chem. 265, 532–537.
Hunter, C. A., and Sanders, J. K. M. (1990).J. Am. Chem. Soc. 112, 5525–5534.
Hunter, C. A., Singh, J., and Thornton, J. M. (1991).J. Mol. Biol. 216, 1–10.
Kanei-Ishii, C., Sarai, A., Sawazaki, T., Nakaghoshi, H., He, D.-N., Ogata, K., Nishimura, Y., and Ishii, S. (1990).J. Biol. Chem. 265, 19990–19995.
Karplus, M., and Petsko, G. A. (1990).Nature (London)347, 631–639.
Kirley, T. L., and Peng, M. (1991).J. Biol. Chem. 266, 19953–19957.
MacLennan, D. H., Brandl, C. J., Korczak, B., and Green, N. M. (1985).Nature (London)316, 696–700.
Mandala, S. M., and Slayman, C. W. (1989).J. Biol. Chem. 264, 16276–16282.
Mata, A. M., Matthews, I., Tunwell, R. E. A., Sharma, R. P., Lee, A. G., and East, J. M. (1992).Biochem. J. 286, 567–580.
Miller, M. J., Oldenburg, M., and Fillingame, R. H. (1990).Proc. Natl. Acad. Sci. USA 87, 4900–4904.
Modyanov, N., Lutsenko, S., Chertova, E., Efremov, R., and Gulyaev, D. (1992).Acta Physiol. Scand. 146, 49–58.
Monk, B. C., Montesinos, C., Ferguson, C., Leonard, K., and Serrano, R. (1991).J. Biol. Chem. 266, 18097–18103.
Munson, K. B., Gutierrez, C., Balaji, V. N., Ramnarayan, K., and Sachs, G. (1991).J. Biol. Chem. 266, 18976–18988.
Na, S., Perlin, D. S., Seto-Young, D., Wang, G., and Haber, J. B. (1993).J. Biol. Chem. 268, 11792–11797.
Nakamoto, R., Rao, R., and Slayman, C. W. (1989).Anal. N.Y. Acad. Sci. 574, 165–179.
Perlin, D. S., Brown, C. L., and Haber, J. E. (1988).J. Biol. Chem. 263, 18118–18122.
Price, E. M., Rice, D. A., and Lingrel, J. B. (1989).J. Biol. Chem. 264, 21902–21906.
Price, E. M., Rice, D. A. and Lingrel, J. B. (1990).J. Biol. Chem. 265, 6638–41.
Rabon, E. C., Bin-Im, W., and Sachs, G. (1988).Methods-Enzymol. 157, 649–54.
Rao, U. S., and Scarborough, G. A. (1990).J. Biol. Chem. 265, 7227–7235.
Rao, U. S., Hennessey, J. P., and Scarborough, G. A. (1991).J. Biol. Chem. 266, 14740–14746.
Richardson, J. S., and Richardson, D. C. (1988).Science 240, 1648–1652.
Sachs, G., Besancon, M., Shin, J. M., Mercier, F., Munson, K., and Hersey, S. (1992).J. Bioenerg. Biomembr. 24, 301–308.
Sali, A., Overington, J. P., Johnson, M. S., and Blundell, T. L. (1990).Trends Biochem. Sci. 15, 235–240.
Serrano, R. (1988).Biochim. Biophys. Acta 947, 1–28.
Serrano, R., and Portillo, F. (1990).Biochem. Biophys. Acta 1081, 195–199.
Serrano, R., Kielland-Brandt, M. C., and Fink, G. R. (1986).Nature (London) 319, 689–693.
Seto-Young, D., and Perlin, D. S. (1991).J. Biol. Chem. 266, 1383–1389.
Seto-Young, D., Monk, B. C., and Perlin, D. S. (1992).Biochim. Biophys. Acta 1102, 213–219.
Shull, G. E. and Lingrel, J. B. (1986).J. Biol. Chem. 261, 16788–16791.
Suga, H., Shirabe, K., Yamamoto, T., Tasumi, M., Umeda, M., Nishimura, C., Nakazawa, A., Nakanishi, M., and Arata, Y. (1991).J. Biol. Chem. 266, 13537–13543.
Thornton, J. M., and Gardner, S. P. (1990). Protein Motifs and Database Searching.Protein: Form and Function, Elsevier Trends Journals, Cambridge, pp. 153–161.
Wach, A., Schlesser, A., and Goffeau, A. (1992).J. Bioenerg. Biomembr. 24, 309–317.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Monk, B.C., Feng, W.C., Marshall, C.J. et al. Modeling a conformationally sensitive region of the membrane sector of the fungal plasma membrane proton pump. J Bioenerg Biomembr 26, 101–115 (1994). https://doi.org/10.1007/BF00763222
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00763222