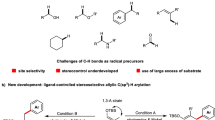
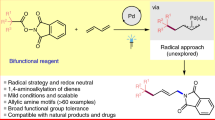
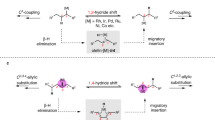
Summary
Allylaromatics undergo isomerization by the use of chlorodicarbonylrhodium(I) dimer as the metal catalyst and phase transfer catalysis conditions [benzene or methylene chloride as the organic phase, aqueous NaOH, and a quarternary ammonium salt as the phase transfer catalyst]. Isomerization of allyl amines and sulfones occurred as well, but the rhodium(I) complex was not required in the latter case.
Similar content being viewed by others
References
E. W. R. Steacie Fellow, 1980–82.
H. Alper and S. Amaratunga,Tetrahedron Lett., 21, 2603 (1980).
D. L. Reger and M. M. Habib,J. Mol. Catal., 7, 365 (1980).
H. Alper and K. Hachem,J. Org. Chem., 45, 2269 (1980) and refs cited therein.
Y. Sasson, A. Zoran, and J. Blum,J. Mol. Catal., 6, 289 (1979).
A. Jonczyk, A. Kwast, and M. Makosza,J. Chem. Soc. Chem. Commun., 902 (1977).
W. E. Truce, T. C. Klingler, and W. W. Brand in S. Oae (Ed.),Organic Chemistry of Sulfur, Plenum Press, New York, 1977, pp. 527–602 and refs cited therein.
Ref. 7, p. 552.
G. Cardillo, D. Savoia, and A. Umani-Ronchi,Synthesis, 453 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alper, H., Hachem, K. Phase transfer and rhodium (I) catalyzed isomerization of allylaromatics and cognate systems. Transition Met Chem 6, 219–220 (1981). https://doi.org/10.1007/BF00618227
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00618227