Summary
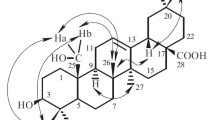
The roots ofFerula lapidosa have yielded a new ester of the carotane series — lapiferin — and its structure and configuration have been established on the basis of chemical transformations and the analysis of spectral characteristics.
Similar content being viewed by others
Literature cited
L. A. Golovina and A. I. Saidkhodzhaev, Khim. Prir. Soedin., 318 (1981).
K. Rajendran, S. K. Paknikar, G. K. Trivedi, and S. C. Bhattacharyya, Indian J. Chem.,16B, 4 (1978).
M. C. Sriraman, B. A. Nagasampagi, R. C. Pandey, and Sukh Dev, Tetrahedron,29, 985 (1972).
A. I. Saidkhodzhaev and G. K. Nikonov, Khim. Prir. Soedin., 559 (1972); 28 (1973); 166 (1974); 105 (1976).
M. Holub, Z. Samek, V. Herout, and F. Sorm, Collect. Czech. Chem. Commun.,32, 591 (1967).
S. M. Kupchan, J. E. Kelsey, M. Maruyama, J. M. Cassady, J. C. Hemingway, and J. R. Knox, J. Org. Chem.,34, 3876 (1969).
N. S. Bhacca and D. H. Williams, Applications of NMR Spectroscopy in Organic Chemistry, Holden-Day, San Francisco (1964).
L. Fieser and M. Fieser, Steroids, Reinhold, New York (1959).
A. A. Savina, D. A. Fesenko, L. I. Dukhovlinova, Yu. E. Sklyar, M. G. Pimenov, and Yu. V. Baranova, Khim. Prir. Soedin., 49 (1979).
A. I. Saidkhodjaev, L. A. Golovina, and V. M. Malikov, 9th Conference on Isoprenoids, Abstracts of Papers, Prague, September (1981).
Additional information
Institute of the Chemistry of Plant Substances of the Academy of Sciences of the Uzbek SSR, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 3, pp. 296–301, May–June, 1983.
Rights and permissions
About this article
Cite this article
Golovina, L.A., Saidkhodzhaev, A.I., Abdullaev, N.D. et al. Structure and stereochemistry of lapiferin. Chem Nat Compd 19, 281–285 (1983). https://doi.org/10.1007/BF00579759
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00579759