Abstract
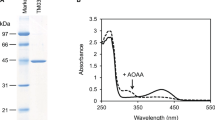
A novel aminotransferase catalyzing the second step of lysine catabolism, the oxidative transamination of the α-group of N6-acetyllysine, was identified and characterized in the yeastCandida maltosa. The enzyme was strongly induced in cells grown on L-lysine as sole carbon source. Its activity was specific for both N6-acetyllysine and 2-oxoglutarate. The Km values were 14 mM for the donor, 4 mM for the acceptor and 1.7 μM for pyridoxal-5-phosphate. The enzyme had a maximum activity at pH 8.1 and 32°C. Its molecular mass estimated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis was 55 kDa. Since the native molecular mass determined by gel filtration was 120 kDa, the enzyme is probably a homodimer.
Similar content being viewed by others
References
Bozler G, Robertson JM, Ohsugi M, Hensley C & Barker HA (1979) Metabolism of L-β-lysine in aPseudomonas: conversion of 6-N-acetyl-L-β-lysine to 3-keto-6-acetamidohexanoate and of 4-aminobutyrate to succinic semialdehyde by different transaminases. Arch. Biochem. Biophys. 197: 226–235
Fothergill JC & Guest JR (1977) Catabolism of L-lysine byPseudomonas aeruginosa. J. Gen. Microbiol. 99: 139–155
Gaillardin C, Fournier P, Sylvestre G & Heslot H (1976) Mutants ofSaccharomycopsis lipolytica defective in lysine catabolism. J. Bacteriol. 125: 48–57
Guengerich FP & Broquist HP (1976) Lysine catabolism inRhizoctonia leguminicola and related fungi. J. Bacteriol. 126: 338–347
Hammer T, Bode R & Birnbaum D (1991a) Occurrence of a novel yeast enzyme, L-lysine ε-dehydrogenase, which catalyses the first step of lysine catabolism inCandida albicans. J. Gen. Microbiol. 137: 711–715
Hammer T, Bode R, Schmidt H & Birnbaum D (1991b) Distribution of three lysine-catabolizing enzymes in various yeast species. J. Basic Microbiol. 31: 43–49
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 227: 680–685
Large PJ & Robertson A (1991) The route of lysine breakdown inCandida tropicalis. FEMS Microbiol. Lett. 82: 209–214
Lowry OH, Rosebrough NJ, Farr AL & Randall RJ (1951) Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275
Rothstein M (1965) Intermediates of lysine dissimilation in the yeast,Hansenula saturnus. Arch. Biochem. Biophys. 111: 467–476
Schmidt H, Bode R & Birnbaum D (1988a) A novel enzyme, L-lysine: pyruvate aminotransferase, catalyses the first step of lysine catabolism inPichia guilliermondii. FEMS Microbiol. Lett. 49: 203–206
Schmidt H, Bode R & Birnbaum D (1988b) Lysine degradation inCandida maltosa: occurrence of a novel enzyme, acetyl-CoA: L-lysine N-acetyltransferase. Arch. Microbiol. 150: 215–218
Tanaka A, Ohishi N & Fukui S (1967) Studies on the formation of vitamins and their function in hydrocarbon fermentation. Production of vitamin B6 byCandida albicans in hydrocarbon medium. J. Ferment. Technol. 45: 617–623
Vogel HJ & Shimura Y (1971) Spectrophotometric determination of lysine. In: Tabor H & Tabor CW (Eds) Methods in Enzymology 17B (pp 228–229). Academic Press, New York/London
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schmidt, H., Bode, R. Characterization of a novel enzyme, N6-acetyl-L-lysine: 2-oxoglutarate aminotransferase, which catalyses the second step of lysine catabolism inCandida maltosa . Antonie van Leeuwenhoek 62, 285–290 (1992). https://doi.org/10.1007/BF00572596
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00572596