Abstract
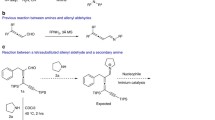
The reaction of 5-ethoxyoxazole with Β-acetylacrylic acid and its ethyl ester is examined, and the most probable mechanism for the heterodiene synthesis with oxazoles is suggested on the basis of the experimental results and on the calculation of the π-electron densities for these molecules.
Similar content being viewed by others
References
G. Ya. Kondrat'eva, Khim. nauka i prom., 2, 666, 1957.
W. Korytnyk and B. Paul, J. Heterocycl. Chem., 2, 481, 1965.
F. Hoffmann-La Roche and Co., A.-G., Neth. Appl. 6506703, 1965; C. A., 64, 15851, 1966.
T. Naito and T. Yoshikawa, Chem. Pharm. Bull., 14, 918, 1966.
Takeda Chemical Industries, Ltd., French patent no. 1400843, 1965; C. A., 63, 9922, 1965.
F. Hoffmann-La Roche and Co., A.-G. French patent no. 1384099, 1965; C. A., 63, 4263, 1965.
T. Yoshikawa, F. Ishikawa, Y. Omura, and T. Naito, Chem. Pharm. Bull., 13, 873, 1965.
J. M. Osbond and F. Hoffman-La Roche and Co., A.-G, French patent no. 1343270, 1963; C. A., 60, 11991, 1964.
H. J. Bernstein and W. G. Schneider, J. Chem. Phys., 24, 469, 1956.
E. N. Dement'eva, N. A. Drobinskaya, L. V. Ionova, M. Ya. Karpeiskii, and V. L. Florent'ev, Biokhimiya, 33, 350, 1968.
W. G. Overend, L. M. Murton, and L. F. Wiggins, J. Chem. Soc., 3500, 1950.
Merck and Co. Inc., Belgian patent no. 617499, 1964.
W. Korytnyk, B. Paul, and A. Bloch, J. Med. Chem., 10, 345, 1967.
C. H. De Puy and K. L. Eilers, J. Org. Chem., 24, 1380, 1959.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Drobinskaya, N.A., Ionova, L.V., Karpeiskii, M.Y. et al. The mechanism of the diene synthesis with 5-alkoxyoxazoles. Chem Heterocycl Compd 6, 33–37 (1970). https://doi.org/10.1007/BF00475419
Issue Date:
DOI: https://doi.org/10.1007/BF00475419