Abstract
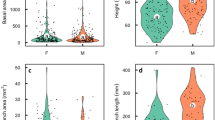
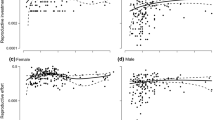
Costs of allocation to male versus female functions were determined for the monecious, annual vine Lagenaria siceraria by removing all flower buds of a given gender and using the additional vegetative growth as a measure of the cost of allocation to that gender (following methods of Silvertown 1987). In this cucurbit, we found significant costs associated with male flower production and fruit production, but not with female flower production. These results are not surprising given the strongly male-biased floral ratio (20 male:1 female) and the large gourds of this species. However, our results are in contrast to Silvertown's study of Cucumis sativus which found no cost to male funtion. In addition, our treatments significantly affected floral sex expression, resulting in increased femaleness for treatment plants. This increase is a consequence of increased lateral branch initiation in these plants and the almost exclusive production of female flowers on lateral branches. Fruit production was associated with a shut-down of subsequent flower production. Given that plant architecture leads to pronounced protandry (on average, 17 male flowers bloom before the first female flower) and that male costs are associated with flower production and the bulk of female costs with fruit production, this shutdown effectively separates male and female functions temporally. We agree with Silvertown's conclusion that this form of protandry is an important factor in the maintenance of cosexuality.
Similar content being viewed by others
References
Ashman T-L (1992) Indirect costs of seed production within and between seasons in a gynodioecious species. Oecologia 92:266–272
Ashman T-L, Baker I (1992) Variation in floral sex allocation with time of season and currency. Ecology 73:1237–1243
Bazzaz FA, Carlson RW (1979) Photosynthetic contribution of flowers and seeds to reproductive effort in an annual colonizer. New Phytol 82:223–232
Bazzaz FA, Carlson RW, Harper JL (1979) Contribution to reproductive effort by photosynthesis of flowers and fruits. Nature 279:554–555
Bierzychudek P (1982) The demography of jack-in-the-pulpit, a forest perennial that changes sex. Ecol Monogr 52:335–351
Broyles SB, Wyatt R (1990) Paternity analysis in a natural population of Asclepias exaltata: multiple paternity, functional gender, and the “pollen donation” hypothesis. Evolution 44:1454–1468
Burd M, Head G (1992) Phenological aspects of male and female function in hermaphroditic plants. Am Nat 140:305–324
Charnov EL (1982) The theory of sex allocation. Princeton University Press, Princeton, NJ
Charnov EL, Maynard Smith J, Bull JJ (1976) Why be an hermaphrodite? Nature 263:125–126
Condon MA, Gilbert LE (1988) Sex expression of Gurania and Psiguria (Cucurbitaceae): neotropical vines that change sex. Am J Bot 75:875–884
Delesalle VA (1987) Patterns of gender allocation in a monoecious cucurbit, Apodanthera undulata, and their reproductive consequences. PhD thesis, University of Arizona, Tucson
Delesalle VA (1992) Architecture and gender allocation: floral sex expression along branches of the monoecious cucurbit, Apodanthera undulata. Int J Plant Sci 153:108–116
Delph LF (1990) Sex-differential resource allocation patterns in the subdioecious shrub Hebe subalpina. Ecology 71:1342–1351
Devlin B, Ellstrand NC (1990) Male and female fertility variation in wild radish, a hermaphrodite. Am Nat 136:87–107
Devlin B, Clegg J, Ellstrand NC (1992) The effect of flower production on male reproductive success in wild radish populations. Evolution 46:1030–1042
Farris MA, Lechowicz MJ (1990) Functional interactions among traits that determine reproductive success in a native annual plant. Ecology 71:548–557
Freeman DC, Klikoff LG, Harper KT (1976) Differential resource utilization by the sexes of dioecious plants. Science 193:597–599
Freeman DC, McArthur ED, Harper KT, Blauer AC (1981) Influence of environment on the floral sex ratio of monoecious plants. Evolution 35:194–197
Goldman DA, Willson MF (1986) Sex allocation in functionally hermaphroditic plants: a review and critique. Bot Rev 52:157–194
Gregg KB (1978) The interaction of light intensity, plant size, and nutrition in sex expression in Cycnoches (Orchidaceae). Selbyana 2:212–223
Jones CS (1992) Comparative ontogeny of a wild cucurbit and its derived cultivar. Evolution 46:1827–1847
Kohn JR (1989) Sex ratio, seed production, biomass allocation, and the cost of male function in Cucurbita foetidissima. Evolution 43:1424–1434
Lloyd DG (1979) Parental strategies of angiosperms. NZ J Bot 17:595–606
Lloyd DG (1980) Sexual strategies in plants. I. An hypothesis of serial adjustment of maternal investment during one reproductive season. New Phytol 86:69–79
Lloyd DG (1982) Selection of combined versus separate sexes in seed plants. Am Nat 120:571–585
Lloyd DG (1984) Gender allocations in outcrossing cosexual plants. In: Dirzo R, Sarukhan J (eds) Perspectives in plant population ecology. Sinauer, Sunderland, pp 273–303
Lloyd DG, Bawa KS (1984) Modification of the gender of seed plants in varying conditions. Evol Biol 17:255–338
Lovett Doust J, Harper JL (1980) The resource costs of gender and maternal support in an andromonoecious umbellifer Smyrnium olusatrum L. New Phytol 85:251–264
Lovett Doust J, Lovett Doust L (1988) Plant reproductive ecology: patterns and strategies. Oxford University Press, Oxford
McKone MJ (1987) Sex allocation and outcrossing rate: a test of theoretical predictions using bromegrasses (Bromus). Evolution 41:591–598
Meagher T (1991) Analysis of paternity with a natural population of Chamaelirium luteum. II. Patterns of male reproductive success. Am Nat 137:738–752
Nitsch J, Kurtz EB, Livermann JL, Went FW (1952) The development of sex expression in Cucurbita flowers. Am J Bot 39:32–43
Policansky D (1981) Sex choice and the size advantage model in jack-in-the-pulpit (Arisaema triphyllum). Proc Natl Acad Sci USA 78:1306–1308
Reckie EG, Bazzaz FA (1987a) Reproductive effort in plants. 1. Carbon allocation to reproduction. Am Nat 129:876–896
Reekie EG, Bazzaz FA (1987b) Reproductive effort in plants. 2. Does carbon reflect the allocation of other resources. Am Nat 129:897–906
Ritland C, Ritland K (1989) Variation of sex allocation among eight taxa of the Mimulus guttatus species complex (Scrophulariaceae). Am J Bot 76:1731–1739
Rodrigues-Pereira AS (1968) Sex expression in floral buds of acorn squash. Planta 80:349–358
Schlessman MA (1988) Gender diphasy. In: Lovett Doust J, Lovett Doust L (eds) Plant reproductive ecology: patterns and strategies. Oxford University Press, Oxford, pp 139–153
Schoen D (1982) Male reproductive effort and breeding system in an hermaphrodite plant. Oecologia 52:255–257
Schoen D, Stewart SC (1986) Variation in male reproductive investment and male reproductive success in white spruce. Evolution 40:1109–1120
Silvertown J (1985) Survival, fecundity and growth of wild cucumber, Echinocystis lobata. J Ecol 73:841–849
Silvertown J (1987) The evolution of hermaphroditism: an experimental test of the resource model. Oecologia 72:157–159
Stanton ML, Bereczky JK, Hasbrouck HD (1987) Pollination thoroughness and maternal yield regulation in wild radish, Raphanus raphanistrum (Brassicaceae). Oecologia 74:68–76
Stephenson AG (1981) Flower and fruit abortion: proximate causes and ultimate functions. Annu Rev Ecol Syst 12:253–279
Stephenson AG (1992) The regulation of maternal investment in plants. In: Marshall C, Grace J (eds) Fruit and seed production: aspects of developmental, environmental physiology and ecology. Cambridge University Press, Cambridge, UK, pp 151–171
Sutherland S (1986) Floral sex ratios, fruit-set, and resource allocation in plants. Ecology 67:991–10001
Sutherland S, Delph LF (1984) On the importance of male fitness in plants: patterns of fruit set. Ecology 65:1093–1104
Waller DM (1988) Plant morphology and reproduction. In: Lovett Doust J, Lovett Doust L (eds) Plant reproductive ecology: patterns and strategies. Oxford University Press, Oxford, pp 203–227
Whitaker TW (1931) Sex ratio and sex expression in the cultivated cucurbits. Am J Bot 18:359–366
Whitaker TW, Davis GN (1962) Cucurbits: botany, cultivation, and utilization. Interscience, New York
Willson MF, Burley N (1983) Mate choice in plants: tactics mechanisms, and consequences. Princeton University Press, Princeton
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Delesalle, V.A., Mooreside, P.D. Estimating the costs of allocation to male and female functions in a monoecious cucurbit, Lagenaria siceraria . Oecologia 102, 9–16 (1995). https://doi.org/10.1007/BF00333304
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00333304