Summary
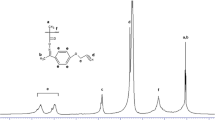
p-(2,3-Dicyano-2,3-dicarbomethoxycyclopropyl)phenyl acrylate 3a and methacrylate 3b were prepared by the reactions of methyl bromocyanoacetate with methyl acryloyloxybenzylidenecyanoacetate 2a or methyl p-methacryloyloxybenzylidenecyanoacetate 2b, respectively. Monomers 3a and 3b were polymerized with free-radical initiators to obtain polymers with multicyanocyclopropane functionalities in the pendant group. The resulting polymers 4a-b were soluble in acetone and the inherent viscosities were in the range of 0.15–0.25 dL/g. Solution-cast films were brittle, showing Tg values in the range of 120–150°C.
Similar content being viewed by others
References
H.K. Hall, Jr., J. Macromol. Sci., Chem., A25, 729 (1988).
Y.S. Jo, Y. Inoue, R. Chujo, K. Saito, and S. Miyata, Macromolecules, 18, 1850 (1985).
G. Henrici-Olive and S. Olive, Adv. Polym. Sci., 6, 421 (1975).
H.K. Hall, Jr., R.T.H. Chan, J. Oku, O.R. Hughes, J. Scheinbeim, and B. Newman, Polym. Bull., 17, 135 (1987).
J.-Y. Lee and H.K. Hall, Jr., Polym. Bull., 23 471 (1990).
J.-Y. Lee, A.B. Padias, and H.K. Hall, Jr., Macromolecules, 24, 17 (1991).
J.-Y. Lee, S.-O. Cho, A.B. Padias, and H.K. Hall, Jr., Polym. Bull., 27, 25 (1991).
A.B. Padias and H.K. Hall, Jr., Polym. Bull., 24 195 (1990).
B.C. Hesse, J. Am. Chem. Soc., 18, 723 (1986).
B.B. Corson and R.W. Stoughton, J. Am. Chem. Soc., 50, 2825 (1928).
Y.C. Kim and H. Hart, J. Chem. Soc. (C), 2409 (1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, JY., Kim, KA., Padias, A.B. et al. Synthesis and radical polymerization of p-(2,3-dicyano-2,3-dicarbomethoxycyclopropyl)phenyl acrylate and methacrylate. Polymer Bulletin 31, 517–522 (1993). https://doi.org/10.1007/BF00297886
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00297886