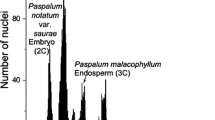
Summary
Mesophyll cell nuclei isolated from leaves of Pennisetum purpureum were analysed by flow cytometry to determine the nuclear DNA content and the percentage of cells in different phases of the cell cycle. Samples taken from base, middle and tip regions of leaves 2 to 8 (leaf 1, which was adjacent to the meristem, was too small to sample) showed no significant differences in the amount of DNA per G1 nucleus due to either age or position. The average amount of DNA per G1 nucleus was 5.78 pg. Although the majority of cells for each sample were in G1, samples taken from older leaves had higher percentages of cells in G2 and S phases. More specifically, base and middle regions of older leaves had a higher percentage of cells in G2 than all three positions in younger leaves. Electrophoretic analysis of nuclear DNA from leaves 2 to 7 showed no evidence of degradation or difference in fragment size for any sample or position. This study was compared to previous work on the relationship between leaf age and embryogenic competence in Pennisetum purpureum. The results suggest that changes in the cell cycle, and/or a loss or fragmentation of the nuclear DNA, are not responsible for loss of embryogenic competence in mature leaf tissue.
Similar content being viewed by others
References
Beaulieu GC, Rogers SO, Bendich AJ (1985) DNA extracted from wheat leaves is highly degraded: a possible basis for the difficulty in establishing leaf cell cultures in the Gramineae. Abstract, 1st Int Congress Plant Mol Biol, Savannah, Georgia
Delllaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol Biol Rep 1:19–23
Fukuda H, Komamine A (1981a) Relationship between tracheary element differentiation and the cell cycle in single cells isolated from the mesophyll of Zinnia elegans. Physiol Plant 52:423–430
Fukuda H, Komamine A (1981b) Relationship between tracheary element differentiation and DNA synthesis in single cells isolated from the mesophyll of Zinnia elegans — analysis by inhibitors of DNA synthesis. Plant Cell Physiol 22:41–49
Galbraith DW (1984) Flow cytometric analysis of the cell cycle. In: Vasil IK (ed) Cell culture and somatic cell genetics of plants, vol 1. Laboratory procedures and their applications. Academic Press, Orlando, pp 765–777
Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP, Firoozabady E (1983) Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220:1049–1051
Gould AR (1983) Control of the cell cycle in cultured plant cells. CRC Crit Rev Plant Sci 1:315–344
Hanning G, Conger BV (1982) Embryoid and plantlet formation from leaf segments of Dactylis glomerata L. Theor Appl Genet 63:155–159
Haydu Z, Vasil IK (1981) Somatic embryogenesis and plant regeneration from leaf tissue and anthers of Pennisetum purpureum Schum. Theor Appl Genet 59:269–273
Hesemann CU, Schröder G (1982) Loss of nuclear DNA in leaves of rye. Theor Appl Genet 62:128–131
Ho W, Vasil IK (1983) Somatic embryogenesis in sugarcane (Saccharum officinarum L.). I. The morphology and physiology of callus formation and the ontogeny of somatic embryos. Protoplasma 118:169–180
Joarder OI, Joarder NH, Dale PJ (1986) In vitro response of leaf tissues from Lolium multiflorum — a comparison with leaf segment position, leaf age and in vivo mitotic activity. Theor Appl Genet 73:286–291
Jones MC, Boffey SA (1984) Deoxyribonuclease activities of wheat seedlings. FEBS Lett 174:215–218
Karlsson SB, Vasil IK (1986) Growth, cytology and flow cytometry of embryogenic cell suspension cultures of Panicum maximum Jacq. and Pennisetum purpureum Schum. J Plant Physiol 123:211–227
Linacero R, Vazquez AM (1986) Somatic embryogenesis and plant regeneration from leaf tissues of rye (Secale cereale L.). Plant Sci 44:219–222
Lu C, Vasil IK (1981) Somatic embryogenesis and plant regeneration from leaf tissue of Panicum maximum Jacq. Theor Appl Genet 59:275–280
Rajasekaran K, Hein MB, Davis GC, Carnes MG, Vasil IK (1987a) Endogenous growth regulators in leaves and tissue cultures of Pennisetum purpureum Schum. J Plant Physiol 130:13–25
Rajasekaran K, Hein MB, Vasil IK (1987b) Endogenous abscisic acid and indole-3-acetic acid and somatic embryogenesis in cultured leaf explants of Pennisetum purpureum Schum.: effects in vivo and in vitro of glyphosate, fluridone and paclobutrazol. Plant Physiol 84:47–51
Rogers SO, Bendich AJ (1985) Extraction of DNA from milligram amounts of fresh, herbarium and mummified plant tissues. Plant Mol Biol 5:69–76
Vasil IK (1987) Developing cell and tissue culture systems for the improvement of cereal and grass crops. J Plant Physiol 128:193–218
Vasil IK, Vasil V (1986) Regeneration in cereal and other grass species. In: Vasil IK (ed) Cell culture and somatic cell genetics of plants, vol 3. Regeneration and variability. Academic Press, Orlando, pp 121–150
Wernicke W, Brettell R (1980) Somatic embryogenesis from Sorghum bicolor leaves. Nature 287:138–139
Wernicke W, Milkovits L (1984) Developmental gradients in wheat leaves — response of leaf segments in different geno-types cultured in vitro. J Plant Physiol 115:49–58
Wernicke W, Milkovits L (1987a) Effect of auxin on the mitotic cell cycle in cultured leaf segments at different stages of development in wheat. Physiol Plant 69:16–22
Wernicke W, Milkovits L (1987b) Rates of uptake and metabolism of indole-3-acetic acid by cultured leaf segments at different stages of development in wheat. Physiol Plant 69:23–28
Wernicke W, Brettell R, Wakizuka T, Potrykus I (1981) Adventitious embryoid and root formation from rice leaves. Z Pflanzenphysiol 103:361–365
Author information
Authors and Affiliations
Additional information
Communicated by G. Wenzel
Rights and permissions
About this article
Cite this article
Taylor, M.G., Vasil, I.K. Analysis of DNA size, content and cell cycle in leaves of Napier grass (Pennisetum purpureum Schum.). Theoret. Appl. Genetics 74, 681–686 (1987). https://doi.org/10.1007/BF00247541
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00247541