Abstract
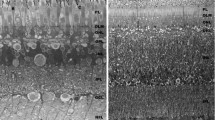
The central retina in hypopigmented mammals is underdeveloped. In the outer retina this deficit is confined to rods. Also, many ganglion cells in temporal regions project inappropriately to the contralateral hemisphere. This study addresses the question of whether pigment-related abnormalities occur in the central retina of a non-mammal, the bird. Birds have a highly developed central retina, but unlike most mammals they do not have a significant uncrossed retinal projection. Consequently, examination of the retinae of hypopigmented birds will reveal whether there is a relationship between the two abnormalities. Also if one of the primary effects of albinism is centred on rods, then albino birds may not show a deficit, because their retinae are cone dominated. Retinae from normally pigmented and two forms of hypopigmented budgerigars (Melopsittacus undulatus) were studied. Measurements of layer thickness, cell density and cell size were made at a range of locations in the ganglion cell layer and in the inner and the outer nuclear layers. Estimates of cone numbers were also made. Each strain of bird had an area of increased retinal layer thickness in dorso-temporal regions, but not a fovea. Although there were variations in the measurements undertaken between the strains, none were pigment related or consistent with the abnormality found in the central retina in albino mammals. Consequently, the underdevelopment of the central retina seen in hypopigmented mammals does not occur in this bird. There are two possible explanations for this result. First, normal mammalian retinal development may depend partly on time-dependent interactions in the maturation of the retinal pigment epithelium and the neural retina. Although there is a common time table for the development of the mammalian visual system when expressed in terms of the caecal period, which is between conception and eye opening, the pace of retinal development in birds is accelerated, which may alter interactions between these regions. Second, as the bird retina is cone dominated, any deficits in albino strains may be relatively minor.
Similar content being viewed by others
References
Baker GE, Reese BE (1993) Chiasmatic course of temporal retinal axons in the developing ferret. J Comp Neurol 330:95–104
Balkema GW, Drager UC (1990) Origins of uncrossed retinofugal projections in normal and hypopigmented mice. Vis Neurosci 4:595–604
Beerman F, Schmid E, Schutz G (1992) Expression of the mouse tyrosinase gene during embryonic development: recapitulation of the temperature regulation in transgenic mice. Proc Natl Acad Sci USA 89:2809–2813
Cooper ML, Pettigrew JD (1979) The retinothalamic pathways in Siamese cats. J Comp Neurol 187:313–348
Drager UC (1985) Birthdates of retinal ganglion cells giving rise to the crossed and the uncrossed optic projections in the mouse. Proc R Soc Lond [Biol] 224:57–77
Drager UC, Olsen JF (1980) Origins of crossed and uncrossed retinal projections in pigmented and albino mice. J Comp Neurol 191:383–412
Dreher B, Robinson SR (1988) Development of the retinofugal pathway in birds and mammals: evidence for a common timetable. Brain Behav Evol 31:369–390
Drenhaus U, Rager G (1992) Organisation of the optic chiasm in the hatched chick. Anat Rec 234:605–617
Ebbesson SOE, Tang D (1965) A method for estimating the number of cells in histological sections. J R Microsc Soc 84:449–464
Ehrlich D (1981) Regional specialization of the chick retina as revealed by the size and density of neurons in the ganglion cell layer. J Comp Neurol 195:643–657
Elschnig A (1913) Zur Anatomie des menschlichen Albinoauges. Graefes Arch Ophthalmol 84:401–419
Guillery RW (1982) The optic chiasm of the vertebrate brain. Contrib Sens Physiol 7:39–72
Guillery RW (1986) Neural abnormalities in albinos. Trends Neurosci 9:364–367
Guillery RW, Updyke BV (1976) Retinofugal pathways in normal and albino axolotls. Brain Res 109:235–244
Guillery RW, Hicky TL, Kaas JH, Felleman DJ, Debruyn EJ, Sparks DL (1984) Abnormal central visual pathways in the brain of an albino green monkey. J Comp Neurol 226:165–183
Hamdi JA, Whitteridge D (1954) The representation of the retina on the optic tectum of the pigeon. Q J Exp Physiol 39:111–119
Hayes BP (1984) Structure of the pigeon retina. Prog Retinal Res 1:197–226
Hodos W, Bessette BB, Macko KA, Weiss SRB (1985) Normative data for pigeon vision. Vision Res 25:1525–1527
Hodos W, Miller RF, Fite KV (1991) Age dependent changes in visual acuity and retinal morphology in pigeons. Vision Res 31:669–677
Jeffery G, Kinsella B (1992) Translaminar deficits in the retinae of albinos. J Comp Neurol 326:637–644
Jeffery G, Darling K, Whitmore A (1994) Melanin and the regulation of mammalian photoreceptor topography. Eur J Neurosci 6:657–667
Kinnear PE, Jay B, Witkop CJ (1985) Albinism. Surv Ophthalmol 30:75–101
La Vail JH, Nixon RA, Sidman RL (1978) Genetic control of retinal ganglion cell projections. J Comp Neurol 182:399–421
Leventhal AG (1982) Morphology and distribution of retinal ganglion cells projecting to different layers of the dorsal lateral geniculate nucleus in normal and Siamese cats. J Neurosci 2:1024–1042
McLoon SC, Lund RD (1982) Transient retinofugal pathways in the developing chick. Exp Brain Res 45:277–284
Meier RE, Mihailovic J, Cuenod M (1974) Thalamic organisation of the retino-thalamic-hyperstriatal pathway in the pigeon (Columba livia). Exp Brain Res 19:351–364
Morgan JE, Hendersen Z, Thompson ID (1987) Retinal decussation patterns in pigmented and albino ferrets. Neuroscience 20:519–535
Müller B, Peichl L (1989) Topography of cones and rods in the tree shrew retina. J Comp Neurol 282:581–594
Oyster CW, Takahashi ES, Fry KR, Lam DMK (1987) Ganglion cell density in albino and pigmented rabbit retinas labelled with a ganglion cell-specific monoclonal antibody. Brain Res 425:25–33
Polyak S (1957) The vertebrate visual system. University of Chicago Press, Chicago
Prada C, Puga J, Perez-Mendez LP, Lopez R, Ramirrez G (1990) Spatial and temporal patterns of neurogenesis in the chick retina. Eur J Neurosci 3:559–569
Rager G, Rager U (1978) System-matching by degeneration. I. A quantitative electron microscope study of the generation and degeneration of retinal ganglion cells in the chick. Exp Brain Res 33:65–78
Robinson SR (1987) Ontogeny of the area centralis in the cat. J Comp Neurol 255:50–67
Robinson SR, Dreher B (1990) The visual pathways of eutherian mammals and marsupials develop according to a common timetable. Brain Behav Evol 36:177–195
Schutte M, Hoskins SG (1993) Ipsilaterally projecting retinal ganglion cells in Xenopus laevis: an HRP study. J Comp Neurol 331:482–494
Stone J, Rowe MH, Champion JE (1978) Retinal abnormalities in the Siamese cat. J Comp Neurol 180:773–782
Thanos S, Bonhoeffer F (1984) Development of the transient ipsilateral retinotectal projection in the chick embryo: a numerical fluorescence-microscopic analysis. J Comp Neurol 224: 407–414
Walls GL (1942) The vertebrate eye and its adaptive radiation. Cranbrook, Michigan
Webster MJ, Rowe MH (1991) Disruption of the developmental timing in the albino rat retina. J Comp Neurol 307:460–474
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jeffery, G., Williams, A. Is abnormal retinal development in albinism only a mammalian problem? Normality of a hypopigmented avian retina. Exp Brain Res 100, 47–57 (1994). https://doi.org/10.1007/BF00227278
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227278