Abstract
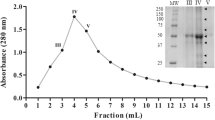
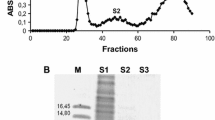
A serine proteinase was isolated from fruits of Maclura pomifera (Raf.) Schneid. by affinity chromatography on bacitracin-containing sorbents and gel-filtration. The enzyme, named macluralisin, is a glycoprotein with a molecular mass of 65 kDa; its protein moiety corresponds to a molecular mass of 50 kDa. The substrate specificity of macluralisin towards synthetic peptides and insulin B-chain is similar to that of cucumisin, a subtilisin-like proteinase from melon fruit. The enzyme is completely inhibited by diisopropylfluorophosphate. Its amino-acid composition resembles that of a serine proteinase isolated from the Cucurbitaceae. The N-terminal sequence has 33% of its residues identical to those of the sequence of fungal subtilisin-like proteinase K. Hence, Maclura pomifera serine proteinase belongs to the subtilisin family, which seems to be broadly distributed in the plant kingdom.
Similar content being viewed by others
Abbreviations
- DFP:
-
diisopropylfluorophosphate
- PMSF:
-
phenylmethylsulfonylfluoride
- Glp:
-
pyroglutamyl
- NHC6H4NO2 :
-
p-nitroanilide
References
Barr, P.J. (1991) Mammalian subtilisins. The long sought dibasic processing endopeptidase. Cell 66, 1–3
Curotto, E., Gonzalez, G. (1989) Isolation and partial characterization of protease from Cucurbita ficifolia. FEBS Lett. 243, 363–365
Filippova, I.Yu., Lysogorskaya, E.N., Oksenoit, E.S., Rudenskaya, G.N., Stepanov, V.M. (1984) l-Pyroglutamyl-l-phenylalanyl-leucine-p-nitroanilide — chromogenic substrate for thiol proteinase assay. Anal. Biochem. 143, 293–297
Jany, K.D., Lederer, G., Mayer, B. (1986) Amino acid sequence of proteinase K from the mold Tritirachium album Limber. FEBS Lett. 199, 139–144
Kaneda, M., Tominago, N. (1975) Isolation and characterization of a proteinase from the sarcocarp of melon fruit. J. Biochem. 78, 1287–1296
Kaneda, M., Tominago, N. (1977) Isolation and characterization of proteinase from Benincasa cerifera. Phytochemistry 16, 345–346
Kaneda, M., Ohmine, H., Yonezawa, H., Tominago, N. (1984) Amino acid sequence around the reactive serine of cucumisin from melon fruit. J. Biochem. 95, 815–829
Kaneda, M., Sobue, A., Eida, S., Tominago, N. (1986) Isolation and characterization of proteinases from the sarcocarp of snake gourd fruit. J. Biochem. 99, 569–577
Kraus, E., Kultw, H.H., Femfert, U.F. (1976) Specificity of proteinase K against oxidized insulin B-chain. Hoppe-Seyler's Z. Physiol. Chem. 357, 233–237
Lobareva, L.S., Rudenskaya, G.N., Stepanov, V.M. (1973) Pepsinlike protease from insectivorous plant Nepenthes. Biokhimiya 38, 642
Lynn, K.R., Clevette-Radford, N.A. (1984) Purification and characterization of hevain, a serine protease from Hevea brasiliensis. Phytochemistry 23, 963–964
Lynn, K.R., Clevette-Radford, N.A. (1986) Ficin E — a serine center protease from Ficus elastica. Phytochemistry 25, 1559–1561
Lynn, K.R., Clevette-Radford, N.A. (1988a) Proteases of Euphorbiaceae. Phytochemistry 27, 45–50
Lynn, K.R., Clevette-Radford, N.A. (1988b) Partenain — a protease from Parthenium argentatum. Phytochemistry 27, 1987–1991
Lyublinskaya, L.A., Haidu, I., Balandina, G.N., Filippova, I.Yu., Markaryan, A.N., Lysogorskaya, E.N., Oksenoit, E.S., Stepanov, V.M. (1987) p-Nitroanilides of pyroglutamyl peptides as chromogenic substrates of serine proteinases. Bioorgan. Khim. 13, 748–753
McGeehan, G., Burkhart, W., Anderegg, R., Becherer, J.D., Gillikin, J.W., Craham, Y.S. (1992) Sequencing and characterization of soybean leaf metalloproteinase. Plant Physiol. 99, 1179–1183
Mosolov, V.V., Loginova, M.D., Malova, E.L., Benken, I.I. (1979) A specific inhibitor of Colletotrichum lindemuthianum protease from kidney bean (Phaseolus vulgaris) seeds. Planta 144, 265–269
Mosolov, V.V., Sokolova, E.V., Livenskaya, O.A. (1984) Inhibitor of chymotrypsin and microbial serine proteinases isolated from corn seeds. Biokhimiya 49, 1334–1342
Rudenskaya, G.N., Stepanov, V.M., Zakharova, Yu.A., Revina, L.P., Khodova, O.M. (1987) A thiol-dependent serine proteinase from sunflower leaves: a bacterial serine proteinase analog. Biokhimiya 52, 1753–1755
Siezen, R.G., Devos, W.M., Leunissen, G.A.M., Dijkstra, B.W. (1991) Homology modelling and protein engineering strategy of subtilases, the family of subtilisin-like serine proteinases. Protein Eng. 4, 719–737
Stepanov, V.M., Rudenskaya, G.N., Gaida, A.V., Osterman, A.L. (1981) Affinity chromatography of proteolytic enzymes on silica-based biospecific sorbents. J. Biochem. Biophys. Methods 5, 177–186
Stepanov, V.M., Rudenskaya, G.N. (1983) Proteinase affinity chromatography on bacitracin-sepharose. J. Appl. Biochem. 5, 420–428
Stepanov, V.M., Rudenskaya, G.N., Vasilyeva, L.I., Krestyanova, I.N., Khodova, O.M., Bartoshevitch, Yu.E. (1986) A serine proteinases from Acremonium chrysogenum. Int. J. Biochem. 18, 369–375
Stepanov, V.M., Rudenskaya, G.N., Revina, L.P., Gryaznova, Yu.B., Lysogorskaya, E.N., Filippova, I.Yu., Ivanova, I.I. (1992) A serine proteinase of an archaebacterium, Halobacterium mediterranei. Biochem. J. 285, 281–286
Tauber, H. (1950) The chemistry and technology of enzymes, pp. 171–172 I. Wiley and Sons, New York
Uchikoba, T., Horita, H., Kaneda, M. (1990) Protease from the sarcocarp of yellow snake-gourd fruit. Phytochemistry 29, 1979–1981
Author information
Authors and Affiliations
Additional information
This work was supported in part by a grant from the Russian Foundation for Basic Research.
Rights and permissions
About this article
Cite this article
Rudenskaya, G.N., Bogdanova, E.A., Revina, L.P. et al. Macluralisin — a serine proteinase from fruits of Maclura pomifera (Raf.) Schneid.. Planta 196, 174–179 (1995). https://doi.org/10.1007/BF00193231
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00193231