Summary
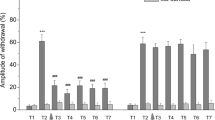
It is possible to obtain habituation of swim induction by stimulating the leech with repetitive light electrical trains. After obtaining this simple form of nonassociative learning, it is also possible to potentiate its response by a series of nociceptive skin brushings (dishabituation). Serotonin applied to the animal is the only neurotransmitter found to mimick dishabituation. We have observed that in the period April–June most animals did not exhibit potentiation of the swimming response after nociceptive stimulation while injection of serotonin mimicked dishabituation as in the animals treated in the period October–March. We have seen correlation between the changes in nonassociative learning and the seasonal variation of serotonin levels in segmental ganglia. This finding strengthens the hypothesis of serotonin as the neurotransmitter mediating dishabituation in swim induction of the leech.
Similar content being viewed by others
Abbreviations
- AHP :
-
afterhyperpolarization
- HPLC :
-
high pressure liquid chromatography
References
Alkon DL, Naito S (1986) Biochemical mechanism of memory storage. J Physiol Paris 81:252–260
Bagnoli P, Magni F (1975) Synaptic inputs to Retzius' cells in the leech. Brain Res 96:147–152
Baylor DA, Nicholls JG (1969) After-effects of nerve impulses on signalling in the central nervous system of the leech. J Physiol Lond 203:571–589
Belardetti F, Biondi C, Colombaioni L, Brunelli M, Trevisani A (1982) Role of serotonin and cAMP on facilitation of the fast conducting system activity in the leech, Hirudo medicinalis. Brain Res 246: 89–103
Belardetti F, Biondi C, Brunelli M, Fabri M, Trevisani A (1983) Heterosynaptic facilitation and behavioral sensitization are inhibited by lowering endogenous cAMP in Aplysia. Brain Res 288:95–104
Belardetti F, Brunelli M, Demontis G, Sonetti D (1984) Serotonin and Retzius cell depress the hyperpolarization following impulses of leech touch cell. Brain Res 300:91–102
Boulis NM, Sahley CL (1988) A behavioral analysis of habituation and sensitization of shortening in the semi-intact leech. J Neurosci 8:4621–4627
Brodfuehrer PD, Friesen WO (1986) Control of leech swimming activity by the cephalic ganglia. J Neurobiol 17:697–705
Brunelli M, Demontis G, Traina G (1985) Role of serotonin and cAMP in short-term changes underlying simple forms of learning process. In: Will BE, Schmitt P, Dalrymple-Alford JC (ed) Brain plasticity, learning and memory. Plenum Publishing Corp., London, pp 97–106
Brunelli M, Colombaioni L, Demontis, Traina G (1986) Role of serotonin on cellular mechanism of non associative learning in swim behavior of the leech Hirudo medicinalis. In: Biggio G, Spano PF, Toffano G, Gessa GL (eds) Modulation of central and peripheral transmitter function. Fidia Res Ser Symp Neurosci III. Liviana Press, Padova, pp 527–531
Brunelli M, Colombaioni L, Traina G (1988) Mechanism of plasticity in short and long-term learning processes in invertebrates. In: Biggio G, Spano PF, Toffano G, Appel SH, Gessa GL (eds) Neuronal plasticity and trophic factors. Liviana Press, Padova, pp 129–132
Coggeshall RE, Yaksta-Sauerland BA (1974) The localization of 5-hydroxytryptamine in chromaffin cells of the leech body wall. J Comp Neurol 156:459–470
Debski EA, Friesen WO (1985) Habituation of swimming activity in the medicinal leech. J Exp Biol 116:169–188
Debski EA, Friesen WO (1986) Role of central interneurons in habituation of swimming activity in the medicinal leech. J Neurophysiol 55:977–994
Debski FA, Friesen WO (1987) Intracellular stimulation of sensory cells elicits swimming activity in the medicinal leech. J Comp Physiol A 160:447–457
Della Corte L, Nistri A (1974) Amine oxidase activity in tissues of the leech Hirudo medicinalis. Br J Pharmacol 52:129P
Dickinson MH, Lent CM (1984) Feeding behavior of the medicinal leech, Hirudo medicinalis. J Comp Physiol A 154:449–455
Kandel ER, Schwartz JH (1982) Molecular biology of learning: modulation of transmitter release. Science 218:433–443
Kandel ER, Klein M, Castellucci VF, Schacher S, Goelet P (1986) Some principles emerging from the study of short- and long-term memory. Neurosci Res 3:498–520
Kilts CD, Breese GR, Mailman RB (1981) Simultaneous quantification of dopamine, 5-hydroxytryptamine and four metabolically related compounds by means of reversed-phase high-performance liquid chromatography with electrochemical detection. J Chromatography 225:347–357
Kristan WB (1983) The neurobiology of swimming in the leech. Trends Neurosci 6:84–88
Kristan WB, McGirr SJ, Simpson GV (1982) Behavioural and mechanosensory neurone responses to skin stimulation in leeches. J Exp Biol 96:143–160
Lent CM (1973) Retzius cells: neuroeffectors controlling mucus release by the leech. Science 179:693–696
Lent CM (1982a) Fluorescent properties of monoamine neurons following glyoxylic acid treatment of intact leech ganglia. Histochemistry 75:77–89
Lent CM (1982b) Serotonin-containing neurones within the segmental nervous system of the leech. In: Osborne NN, Wiley J (eds) Biology of serotonergic transmission. New York, pp 431–455
Lent CM (1984) Quantitative effects of a neurotoxin upon serotonin levels within tissue compartments of the medicinal leech. J Neurobiol 15:309–323
Lent CM, Dickinson MH (1984) Serotonin integrates the feeding behavior of the medicinal leech. J Comp Physiol A 154:457–471
Lent CM, Frazer BM (1977) Connectivity of the monoamine-containing neurones in the central nervous system of the leech. Nature 266:844–847
Mason A, Kristan WB (1982) Neuronal excitation, inhibition and modulation of leech longitudinal muscle. J Comp Physiol 146:527–536
Muller KJ, Nicholls JG, Stent G (eds) (1981) Neurobiology of the leech. Cold Spring Harbor Laboratory, New York
Nusbaum MP, Kristan WB (1986) Swim initiation in the leech by serotonin containing interneurones, cells 21 and 61. J Exp Biol 122:277–302
Nusbaum MP, Friesen WO, Kristan WB, Pearce RA (1987) Neural mechanisms generating the leech swimming rhythm: swim-initiator neurons excite the network of swim oscillator neurons. J Comp Physiol A 161:355–366
Pearce RA, Friesen WO (1985a) Intersegmental coordination of the leech swimming rhythm. I. Roles of cyclic period gradient and coupling strength. J Neurophysiol 54:1444–1459
Pearce RA, Friesen WO (1985b) Intersegmental coordination of the leech swimming rhythm. II. Comparison of long and short chains of ganglia. J Neurophysiol 54:1460–1472
Sahley CL, Ready DF (1988) Associative learning modifies two behaviors in the leech Hirudo medicinalis. J Neurosci 8:4612–4620
Sawyer RT (1986) Leech biology and behaviour. Clarendon Press, Oxford
Stent GS, Kristan WB (1981) The swimming rhythm. In: Muller KJ, Nicholls JG, Stent GS (eds) Neurobiology of the leech. Cold Spring Harbor Laboratory, New York, pp 113–146
Stenzel K, Neuhoff V (1976) Tryptophan metabolism and the occurrence of amino acids and serotonin in the leech (Hirudo medicinalis) nervous system. J Neurosci Res 2:1–9
Stern-Tomlinson W, Nusbaum MP, Perez LE, Kristan WB (1986) A kinematic study of crawling behavior in the leech Hirudo medicinalis. J Comp Physiol A 158:593–603
Willard AL (1981) Effects of serotonin on the generation of the motor program for swimming by the medicinal leech. J Neurosci 1:936–944
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Catarsi, S., Garcia-Gil, M., Traina, G. et al. Seasonal variation of serotonin content and nonassociative learning of swim induction in the leech Hirudo medicinalis . J Comp Physiol A 167, 469–474 (1990). https://doi.org/10.1007/BF00190817
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00190817